��Ŀ����
ʵ���������Ȼ�þ�������ƵĹ���������Ʒ��С��ͬѧ��ⶨ��Ʒ���Ȼ�þ�������������ȳ�ȡ�û������Ʒ20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ
100g���Ĵ����μ������У������ʵ���������ݼ��±�����������������йؼ��㣺
| �������ʵ����� | 1 | 2 | 3 | 4 |
| ��������������Һ������/g | 25 | 25 | 25 | 25 |
| ���ɳ���������/g | 2.9 | X | 8.7 | 8.7 |
��2����������ʵ�������õ�������������Һ����������������
��3������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�
���𰸡���������1���ɱ��е����ݿ�֪ÿ����25g����������Һ��ȫ��Ӧ�����ɳ���2.9g����ˣ���2�μ����25g����������Һ��ȫ��Ӧ�������ɳ���2.9g��
��2������ÿ����2.9g��������������������Һ��������25g�����û�ѧ����ʽ�ɼ��㷴Ӧ�����������������������������������Һ����������������
��2�����Ȼ�þ�����������������ȫ��Ӧ�����ɳ���������þ8.7g�����ݷ�Ӧ�Ļ�ѧ����ʽ���������Ʒ���Ȼ�þ��������Ȼ������Ʒ������ȿɵû������Ʒ���Ȼ�þ������������
����⣺��1��ÿ����25g����������Һ��ȫ��Ӧ�����ɳ���2.9g����ˣ���2�μ���25g����������Һ��ȫ��Ӧ�����ó�����������=2.9g×2=5.8g��
���������Ʒ���Ȼ�þ������Ϊx����֮��Ӧ��������������Ϊy
MgCl2+2NaOH�TMg��OH��2��+2NaCl
95 80 58
x y 8.7g
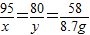
x=14.25g y=12g
��2���ɷ�����֪ÿ����2.9g������þ��Ҫ����������Һ������25g����������8.7g�������������������Һ����Ϊ��25g×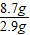 =75g����������ʵ�������õ�������������Һ��������������Ϊ��
=75g����������ʵ�������õ�������������Һ��������������Ϊ��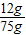 ×100%=16%��
×100%=16%��
��3��ԭ����������Ʒ���Ȼ�þ����������=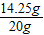 ×100%=71.25%
×100%=71.25%
�ʴ�Ϊ����1��5.8g����2������ʵ�������õ�������������Һ��������������Ϊ16%����3��ԭ����������Ʒ���Ȼ�þ����������Ϊ71.25%��
������������һ��ͼ�����ݷ����ۺ��⣬����Ĺؼ���Ū���ױ��������ݵ����壬�����жϳ���Ӧʱ�����ʵ���������������ۺ��Խ�ǿ���ѶȽϴ�
��2������ÿ����2.9g��������������������Һ��������25g�����û�ѧ����ʽ�ɼ��㷴Ӧ�����������������������������������Һ����������������
��2�����Ȼ�þ�����������������ȫ��Ӧ�����ɳ���������þ8.7g�����ݷ�Ӧ�Ļ�ѧ����ʽ���������Ʒ���Ȼ�þ��������Ȼ������Ʒ������ȿɵû������Ʒ���Ȼ�þ������������
����⣺��1��ÿ����25g����������Һ��ȫ��Ӧ�����ɳ���2.9g����ˣ���2�μ���25g����������Һ��ȫ��Ӧ�����ó�����������=2.9g×2=5.8g��
���������Ʒ���Ȼ�þ������Ϊx����֮��Ӧ��������������Ϊy
MgCl2+2NaOH�TMg��OH��2��+2NaCl
95 80 58
x y 8.7g
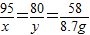
x=14.25g y=12g
��2���ɷ�����֪ÿ����2.9g������þ��Ҫ����������Һ������25g����������8.7g�������������������Һ����Ϊ��25g×
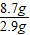 =75g����������ʵ�������õ�������������Һ��������������Ϊ��
=75g����������ʵ�������õ�������������Һ��������������Ϊ��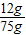 ×100%=16%��
×100%=16%����3��ԭ����������Ʒ���Ȼ�þ����������=
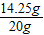 ×100%=71.25%
×100%=71.25%�ʴ�Ϊ����1��5.8g����2������ʵ�������õ�������������Һ��������������Ϊ16%����3��ԭ����������Ʒ���Ȼ�þ����������Ϊ71.25%��
������������һ��ͼ�����ݷ����ۺ��⣬����Ĺؼ���Ū���ױ��������ݵ����壬�����жϳ���Ӧʱ�����ʵ���������������ۺ��Խ�ǿ���ѶȽϴ�

��ϰ��ϵ�д�
 һ����������ϵ�д�
һ����������ϵ�д�
�����Ŀ
a��ʵ���������Ȼ�þ�������ƵĹ���������Ʒ��С��ͬѧ��ⶨ��Ʒ���Ȼ�þ�������������ȳ�ȡ�û������Ʒ20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ100gƽ�����Ĵμ������У������ʵ���������ݼ��±�����������������йؼ��㣺
��1���ϱ���X����ֵΪ ��
��2������20g����������Ʒ���Ȼ�þ��������
��3����������ʵ�������õ�������������Һ����������������
b�����ձ��м����Ȼ��ƺ�̼�����ƵĹ�������10.0g���ټ���68.9gϡ����ǡ����ȫ��Ӧ��
��Ӧ�������þ�����������ձ���ͬҩƷ����������m���뷴Ӧʱ�䣨t���Ĺ�ϵ��ͼ��ʾ���ձ���ͬҩƷ����ʼ������Ϊ165.0g����Ӧ�ķ���ʽΪNaHCO3+HCl=NaCl+H2O+CO2��������
��1����ȫ��Ӧʱ����������̼�������� ��
��2������ԭ��������Ȼ��Ƶ�������
��3������ϡ�������������������
| �������ʵ����� | 1 | 2 | 3 | 4 |
| ��������������Һ������/g | 25 | 25 | 25 | 25 |
| ���ɳ���������/g | 2.9 | X | 8.7 | 8.7 |
��2������20g����������Ʒ���Ȼ�þ��������

��3����������ʵ�������õ�������������Һ����������������
b�����ձ��м����Ȼ��ƺ�̼�����ƵĹ�������10.0g���ټ���68.9gϡ����ǡ����ȫ��Ӧ��
��Ӧ�������þ�����������ձ���ͬҩƷ����������m���뷴Ӧʱ�䣨t���Ĺ�ϵ��ͼ��ʾ���ձ���ͬҩƷ����ʼ������Ϊ165.0g����Ӧ�ķ���ʽΪNaHCO3+HCl=NaCl+H2O+CO2��������
��1����ȫ��Ӧʱ����������̼��������
��2������ԭ��������Ȼ��Ƶ�������
��3������ϡ�������������������
ʵ���������Ȼ�þ�������ƵĹ���������Ʒ��С��ͬѧ��ⶨ��Ʒ���Ȼ�þ�������������ȳ�ȡ�û������Ʒ20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ100gƽ�����Ĵμ������У������ʵ���������ݼ��±�����������������йؼ��㣺
��1���ϱ���X����ֵΪ ��
��2������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�
��3������ʵ�������õ�������������Һ����С��ͬѧ����ʵ���������е�80g������������Ϊ30%������������Һ�����Ƶģ��Լ����������ٿ�ˮ���������ʵ��������������������������������Һ��
| �������ʵ����� | 1 | 2 | 3 | 4 |
| ��������������Һ������/g | 25 | 25 | 25 | 25 |
| ���ɳ���������/g | 2.9 | X | 8.7 | 8.7 |
��2������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�
��3������ʵ�������õ�������������Һ����С��ͬѧ����ʵ���������е�80g������������Ϊ30%������������Һ�����Ƶģ��Լ����������ٿ�ˮ���������ʵ��������������������������������Һ��
ʵ���������Ȼ�þ�������ƵĹ���������Ʒ��С��ͬѧ��ⶨ��Ʒ���Ȼ�þ�������������ȳ�ȡ�û������Ʒ20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ100 gƽ�����Ĵμ������У������ʵ���������ݼ��±�����������������йؼ��㣺
��1���ϱ���X����ֵΪ ��
��2������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�
| �� �� ���ʵ����� |
1 | 2 | 3 | 4 |
| ��������������Һ������/g | 25 | 25 | 25 | 25 |
| ���ɳ���������/g | 2.9 | X | 8.7 | 8.7 |
��2������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�
ʵ���������Ȼ�þ�������ƵĹ���������Ʒ��С��ͬѧ��ⶨ��Ʒ���Ȼ�þ�������������ȳ�ȡ�û������Ʒ20g����ȫ����ˮ�У�Ȼ��ȡ����һ��������������������������Һ100gƽ�����Ĵμ������У������ʵ���������ݼ��±�����������������йؼ��㣺
|
| 1 | 2 | 3 | 4 |
| ��������������Һ������/g | 25 | 25 | 25 | 25 |
| ���ɳ���������/g | 2.9 | X | 8.7 | 8.7 |
��1���ϱ���X����ֵΪ_________��
��2������ԭ����������Ʒ���Ȼ�þ�����������Ƕ��٣�����������ػ�ѧ����ʽ���м��㣬д����Ҫ�Ĺ��̣�
��3������ʵ�������õ�������������Һ����С��ͬѧ����ʵ���������е�80g������������Ϊ30%������������Һ�����Ƶģ�ͨ�������֪����� gˮ���������ʵ��������������������������������Һ��
