��Ŀ����
�����Ͼ����в����̷��Ի�ͭð��ƽ�Ϊ�� �ⶨ��ͭ(ͭ��п�Ͻ�)����ɣ�С�������о���
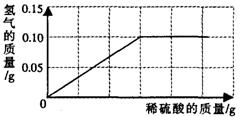
�ⶨ��ͭ(ͭ��п�Ͻ�)����ɣ�С�������о��� ѧϰС���ȡ����Ʒ10g����������μ���9��8����ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ��
ѧϰС���ȡ����Ʒ10g����������μ���9��8����ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ��
�Լ��㣺�����ռ���������һλС����
��1����Ʒ��ͭ������������
��2���� Ӧ��������Һ�����ʵ�����������
Ӧ��������Һ�����ʵ�����������
��3��ͨ������о����㷢���˼����ͭ��ƽ�ķ����� ��
�⣺��п����Ϊx�� ZnSO4����Ϊy��
Zn + H2SO4══ ZnSO4 + H2�� ��2�֣�
65 161 2
x y 0.1g
65:2=X:0.1g x=3.25g ��2�֣�
65:161=y:0.1g y=8.05g  ��2�֣�
��2�֣�
(1)����ͭ��Cu����������Ϊ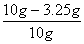 ��100%=66.7% ��2�֣�
��100%=66.7% ��2�֣�
(2) 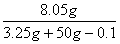 ��100%=15.1% ��2�֣�
��100%=15.1% ��2�֣�
��3�� �μ�ϡ���ᣬ���Ƿ������ݲ����� ��2�֣�
���ԡ�
 |

 ��У����ϵ�д�
��У����ϵ�д�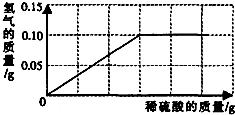 �����Ͼ����в����̷��Ի�ͭð��ƽ�Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�С�������о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ��ͼ��ʾ��
�����Ͼ����в����̷��Ի�ͭð��ƽ�Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�С�������о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ��ͼ��ʾ��