��Ŀ����
ij��ѧ��ȤС�����������ͼ��ʾʵ��װ�á�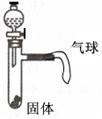
�� ��Һ©�����������е�Һ�岻�����£����ܵ�ԭ���� ��
�� ��������������ʹ���е���ɫҺ�����Թ��еĹ���Ӵ���Ӧ���ɹ۲쵽�����ʹ�������ֱ�д��һ������ͼ�����������Ҫ��Ļ�ѧ����ʽ��
�٣��ֽⷴӦ ��
�ڣ����Ϸ�Ӧ�� �������ʹ��ԭ�� ��
�� ����Һ©����Һ��Ϊϡ���ᣬ����������Щ���������������ᷢ����ѧ��Ӧ����ʹ�����ʹ���� ������ţ��� ��Na2CO3 ��Zn ��Cu ��NaCl
| A���٢� | B���٢ڢ� | C���٢ڢ� | D���٢ڢۢ� |
���� ��Һ©��ƿ��δ��
�� �� 2H2O2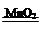
 2H2O+O2�� �� CaO+H2O ==Ca(OH)2��
2H2O+O2�� �� CaO+H2O ==Ca(OH)2��
��������ˮ��Ӧ���ȣ�ƿ������������͡�
�� A ������ʽ2�֣�����ÿ��1�֣�
����

��ϰ��ϵ�д�
 ���ݼ���ϵ�д�
���ݼ���ϵ�д�
�����Ŀ



 ij��ѧ��ȤС�������һ����ʵ��װ�ã������ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ����������������������ǣ�
ij��ѧ��ȤС�������һ����ʵ��װ�ã������ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ����������������������ǣ�