��Ŀ����
����������������������ֻ����ӽǡ�
ˮ��ͨ��������¿��Է�����ѧ��Ӧ�����������ͼ1��ͼ2�ش����⡣


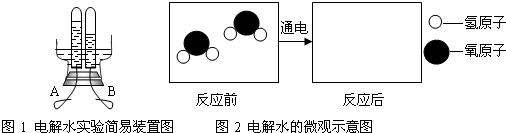
ͼ1��ͬѧ����Ƶĵ��ˮ����װ�ã�����A��B�缫�ɽ����������Ƴɡ�ͨ��
һ��ʱ���A��B�������Թ��в�������������Ϊ �����ˮ�Ļ�ѧ����ʽΪ ����ʵ��˵��ˮ���� ��ɵġ�
��1�����������������ʣ���Ҫ���д��һ����ˮ�йصĻ�ѧ����ʽ
��ѡ���ʣ�������̼�������ơ����������顢����
�� ��ˮ�μӵĻ��Ϸ�Ӧ����������������������������������������������
�� ��ˮ�����Ļ�ѧ��Ӧ����������������������������������������������
��3��ʯ��ׯ��������ȱˮ�ij��У�����ӷ�ֹˮ��Ⱦ����Լ��ˮ�ȷ���д��һ���Լ�
�����ܼ��ľ�����������������������������������������������������
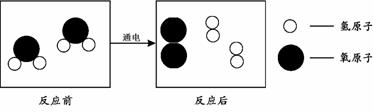
��4������ͼ2���á� ������
������ ����ʾ��Ӧ��������
����ʾ��Ӧ��������
��5���������������������������ͼ2��˵����ѧ��Ӧ�������غ��ԭ���ǣ�ԭ�ӵ�����䡢 ���䡢ԭ�ӵ��������䡣
��1��1��2 2H2O 2H2�� + O2�� �⡢������Ԫ��
2H2�� + O2�� �⡢������Ԫ��
��2���� CaO��H2O=Ca(OH) 2 (����CO2��H2O=H2CO3)
�� 2H2��O2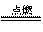 2H2O (����CH4��2O2
2H2O (����CH4��2O2 CO2��2H2O)
CO2��2H2O)
(3)����
(4)
(5) ԭ����Ŀ

 ��
�� ����ʾ��Ӧ��������
����ʾ��Ӧ��������

 ������
������ ����ʾ��Ӧ��������
����ʾ��Ӧ��������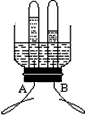

 ������
������ ����ʾ��Ӧ��������
����ʾ��Ӧ��������