��Ŀ����
����С���ͬѧ�ڲⶨ��NaCl��Na2CO3�γɵĹ���������ɵģ�����������ʵ�飺ȡ40g�������ƽ����Ϊ�ķݷ����ĸ������У�Ȼ��ֱ����ͬ��ϡ���ᣬ�����ɶ�����̼�����ռ��������ʵ�����ݼ��±���
| ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
| ԭ������������ | 10g | 10g | 10g | 10g |
| ����ϡ��������� | 10g | 20g | 30g | 40g |
| ���ɶ�����̼������ | 0.88g | m | 2.2g | 2.2g |
������������ݻش��㣨��3С��Ҫ��д��������̣�
��1��10gԭ����������ɵ���Һ������ϡ���ᷴӦ��������ɶ�����̼���������Ϊ______g��
��2��mΪ______g��
��3��10g����������Na2CO3�������Ƕ��٣�
�⣺��1�����ڼ���30g��40g�������ɵĶ�����̼������Ϊ2.2g�����Կ����ж�������ɶ�����̼���������Ϊ2.2g��
��2������ʵ�����ݣ�����10g������ȫ��Ӧ���������������Ϊ0.88g������30g������Һʱ�����������ɣ����жϱ���m��ֵΪ0.88g��2=1.76g��
��3���⣺��10g����������Na2CO3��������x
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 44
x 2.2g

x=5.3g
�ʴ�Ϊ����1��2.2����2��1.76����3��10g����������Na2CO3��������5.3g��
��������1������ʵ�������������ɵĶ�����̼���������ж�������ɶ�����̼�����������
��2������ʵ�����ݣ�����10g������ȫ��Ӧ���������������Ϊ0.88g������30g������Һʱ�����������ɣ����жϱ���m��ֵ��
������������ȫ��Ӧʱ���������������Ϊ2.2g������̼���������ᷴӦ�Ļ�ѧ����ʽ���������ɵĶ�����̼������������ԭ����������Na2CO3��������
������������Ҫ����ѧ�������ݷ����ʹ�������������Ϥ��ȫ��Ӧ������ȫ��Ӧ�ĸ�����ܾݴ˲������ݣ����м����ǽ���Ĺؼ���
��2������ʵ�����ݣ�����10g������ȫ��Ӧ���������������Ϊ0.88g������30g������Һʱ�����������ɣ����жϱ���m��ֵΪ0.88g��2=1.76g��
��3���⣺��10g����������Na2CO3��������x
Na2CO3+2HCl�T2NaCl+H2O+CO2��
106 44
x 2.2g

x=5.3g
�ʴ�Ϊ����1��2.2����2��1.76����3��10g����������Na2CO3��������5.3g��
��������1������ʵ�������������ɵĶ�����̼���������ж�������ɶ�����̼�����������
��2������ʵ�����ݣ�����10g������ȫ��Ӧ���������������Ϊ0.88g������30g������Һʱ�����������ɣ����жϱ���m��ֵ��
������������ȫ��Ӧʱ���������������Ϊ2.2g������̼���������ᷴӦ�Ļ�ѧ����ʽ���������ɵĶ�����̼������������ԭ����������Na2CO3��������
������������Ҫ����ѧ�������ݷ����ʹ�������������Ϥ��ȫ��Ӧ������ȫ��Ӧ�ĸ�����ܾݴ˲������ݣ����м����ǽ���Ĺؼ���

��ϰ��ϵ�д�
 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д� Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д�
�����Ŀ
����С���ͬѧ�ڲⶨ��NaCl��Na2CO3�γɵĹ����������ʱ������������ʵ�飺ȡ40g�������������Һ��ƽ����Ϊ�ķݣ�Ȼ��ֱ����һ������������CaCl2��Һ��ʵ�����ݼ��±���
������������ݻش��㣮
��1��m= g��
��2��ԭ����������Na2CO3�����������Ƕ��٣� ��
| ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
| ԭ������������ | 10g | 10g | 10g | 10g |
| ����CaCl2��Һ���� | 10g | 20g | 30g | 40g |
| ���ɵij��������� | 2g | m | 5g | 5g |
��1��m=
��2��ԭ����������Na2CO3�����������Ƕ��٣�
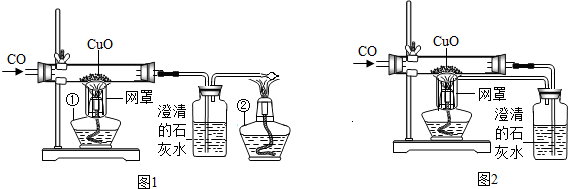
 ijУ����С���ͬѧ����ʦ�İ����£�̽���ó�������Ҫ�ɷ�Fe2O3����������Ҫ��Ӧԭ����������Ƶ�ʵ��װ�ã���ͼ��
ijУ����С���ͬѧ����ʦ�İ����£�̽���ó�������Ҫ�ɷ�Fe2O3����������Ҫ��Ӧԭ����������Ƶ�ʵ��װ�ã���ͼ��