��Ŀ����
�����ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺��1��������֯���˹��������������ʿ������˹�������� ������ţ���
��ʳ�� �ڸɱ� ����� ��ľ̿��
��2����Щ����ȡ���ǵĺ�ˮ��������ˮ�������ǵĺ�ˮ����ͼ��ʾ�ļ���ˮ��
���о���������̿���������Ե�ԭ���� ��
�ô�װ�þ�����õ���ˮ���� ����������������
��3����Щ���ȡ�õ���ˮ������ij����ˮ��Ӳˮ������ˮ�����õ������� ��
��4��ClO2����һ������ˮ��������������ˮ����������������Cl2��������ˮ����������ȡClO2�ķ�Ӧ
����ʾ��ͼ���£��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��5��ˮ�����츣���࣬��ˮ����Ⱦ��ȴ������������ѣ�Ϊ�˷�ֹˮ����Ⱦ�����и��������ˮ�����ж���ֲ����������ڲ������ŷŹ�ҵ��ˮ���۽�ֹʹ��ũҩ�ͻ��ʣ���������ˮ�����������������ŷţ����п��Բ��õķ����� ������ĸ����
A���٢�B���ڢ�C���٢�D���ڢ�
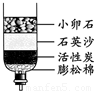
���𰸡���������1���ɱ����������˹����ꣻ
��2������̿�����������ã��ܹ�����ˮ�е�ɫ�غ���ζ��ͨ�����ˡ������ȷ������ܳ�ȥ����ˮ�����ʣ�
��3���÷���ˮ���Լ���ˮ��Ӳ�ȣ�
��4�����ݷ�Ӧ����������Լ���Ӧ�������н��
��5��Ҫ�������ˮ����Ҫ�ԣ�
����⣺��1���ɱ����������˹����꣮����ڣ�
��2�������ǵĺ�ˮͨ������ˮ�����о��������л���̿�Ľṹ�����ɶ�ף�����������ˮ�е�ɫ�غ���ζ���ô�װ�þ�����õ���ˮ����Ȼ����һЩ����ˮ�����ʣ����ڻ���������ɶ�ף�����
��3����ˮ�м������ˮʱ�����������������ĭ��������ˮ�������������ĭ���٣�����Ӳˮ���������ˮ��
��4��NaClO2��Cl2��Ӧ�����Ȼ��ƺͶ������ȣ���Ӧ�Ļ�ѧ����ʽΪ2NaClO2+Cl2=2NaCl+2ClO2�����2NaClO2+Cl2=2NaCl+2ClO2��
��5������ˮ�����ж���ֲ�����������ֹʹ��ũҩ�ͻ��ʵȴ�ʩ���Dz���ʵ�ģ��������ŷŹ�ҵ��ˮ��������ˮ�����������������ŷſ��Ա���ˮ��Դ�����D��
�����������Ҫ���վ���ˮ�ķ����ͽ�Լ��ˮ����Ҫ�ԣ�ֻ���������ܶ���ط��������������ȷ���жϣ�
��2������̿�����������ã��ܹ�����ˮ�е�ɫ�غ���ζ��ͨ�����ˡ������ȷ������ܳ�ȥ����ˮ�����ʣ�
��3���÷���ˮ���Լ���ˮ��Ӳ�ȣ�
��4�����ݷ�Ӧ����������Լ���Ӧ�������н��
��5��Ҫ�������ˮ����Ҫ�ԣ�
����⣺��1���ɱ����������˹����꣮����ڣ�
��2�������ǵĺ�ˮͨ������ˮ�����о��������л���̿�Ľṹ�����ɶ�ף�����������ˮ�е�ɫ�غ���ζ���ô�װ�þ�����õ���ˮ����Ȼ����һЩ����ˮ�����ʣ����ڻ���������ɶ�ף�����
��3����ˮ�м������ˮʱ�����������������ĭ��������ˮ�������������ĭ���٣�����Ӳˮ���������ˮ��
��4��NaClO2��Cl2��Ӧ�����Ȼ��ƺͶ������ȣ���Ӧ�Ļ�ѧ����ʽΪ2NaClO2+Cl2=2NaCl+2ClO2�����2NaClO2+Cl2=2NaCl+2ClO2��
��5������ˮ�����ж���ֲ�����������ֹʹ��ũҩ�ͻ��ʵȴ�ʩ���Dz���ʵ�ģ��������ŷŹ�ҵ��ˮ��������ˮ�����������������ŷſ��Ա���ˮ��Դ�����D��
�����������Ҫ���վ���ˮ�ķ����ͽ�Լ��ˮ����Ҫ�ԣ�ֻ���������ܶ���ط��������������ȷ���жϣ�

��ϰ��ϵ�д�
�����Ŀ
 ��3����ͼ���þ�ˮ��Ͱ������оʵ��ͼ���������ѧ֪ʶ�ش��������⣮
��3����ͼ���þ�ˮ��Ͱ������оʵ��ͼ���������ѧ֪ʶ�ش��������⣮ 23�������ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺
23�������ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺ �����ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺
�����ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺ �����ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺
�����ҹ����ϵ����������أ�ˮ��Դ�ı����ͺ����������ܵ����ǵ��ձ��ע���������������������й����⣺