��Ŀ����
�������۹ܵ��г���ۼ��ж���������������¹ʡ�ijѧϰС����֪���������۹ܵ����ijɷ���ʲô������ͨ���������ϵ�֪��
I���������۹ܵ��п��ܺ��нϴ�����CO��CO2��H2S��CH4�����塣
��H2S�����о綾������CuSO4��Һ��Ӧ���ɺ�ɫ������
��CO2�ᱻNaOH��Һ���գ�CH4��CO������CuSO4��Һ��ʯ��ˮ�Լ�NaOH��Һ��Ӧ��
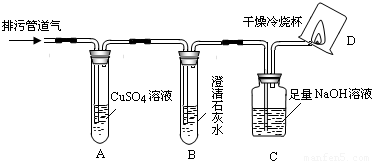
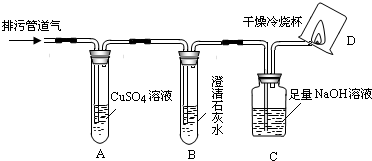
��С���������ͼ��ʾ��װ�ò�����̽��(�г�������ʡ��)��
I���������۹ܵ��п��ܺ��нϴ�����CO��CO2��H2S��CH4�����塣
��H2S�����о綾������CuSO4��Һ��Ӧ���ɺ�ɫ������
��CO2�ᱻNaOH��Һ���գ�CH4��CO������CuSO4��Һ��ʯ��ˮ�Լ�NaOH��Һ��Ӧ��
��С���������ͼ��ʾ��װ�ò�����̽��(�г�������ʡ��)��

��ش��������⣺
��1��װ��A�г��ֺ�ɫ���ǣ�˵�����۹ܵ����к���____________���ѧʽ����
��2��װ��B�г��ְ�ɫ���ǣ�˵�����۹ܵ����к���________________���ѧʽ�����˷�Ӧ�Ļ�ѧ����ʽΪ_________________________________��
��3��װ��D�пɼ����棬������ձ��ڱ���ˮ�����֣�˵�����۹ܵ����к���____________���ѧʽ�����˷�Ӧ�Ļ�ѧ����ʽΪ__________________________����Ҫ��֤������ȼ�պ����һ�ֲ���ɽ��еIJ����ǣ�Ѹ�ٰ��ձ������������ձ���ע��_________________����
��4����ͬѧ��������ȼ�ղ�����ˮ�Ͷ�����̼�����������㣻��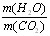 <____________����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO���밴��˼·���������������ա�
<____________����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO���밴��˼·���������������ա�
��1��װ��A�г��ֺ�ɫ���ǣ�˵�����۹ܵ����к���____________���ѧʽ����
��2��װ��B�г��ְ�ɫ���ǣ�˵�����۹ܵ����к���________________���ѧʽ�����˷�Ӧ�Ļ�ѧ����ʽΪ_________________________________��
��3��װ��D�пɼ����棬������ձ��ڱ���ˮ�����֣�˵�����۹ܵ����к���____________���ѧʽ�����˷�Ӧ�Ļ�ѧ����ʽΪ__________________________����Ҫ��֤������ȼ�պ����һ�ֲ���ɽ��еIJ����ǣ�Ѹ�ٰ��ձ������������ձ���ע��_________________����
��4����ͬѧ��������ȼ�ղ�����ˮ�Ͷ�����̼�����������㣻��
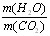 <____________����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO���밴��˼·���������������ա�
<____________����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO���밴��˼·���������������ա� (1)H2S
(2)CO2��CO2+Ca(OH)2==CaCO3��+H2O
(3)CH4��CH4+2O2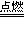 CO2+2H2O������ʯ��ˮ
CO2+2H2O������ʯ��ˮ
(4)9:11
(2)CO2��CO2+Ca(OH)2==CaCO3��+H2O
(3)CH4��CH4+2O2
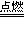 CO2+2H2O������ʯ��ˮ
CO2+2H2O������ʯ��ˮ(4)9:11

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ

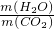 ��______����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO�����밴��˼·���������������գ���
��______����ԭ��Ʒ���к���CO������ԭ��Ʒ���оͲ���CO�����밴��˼·���������������գ���