��Ŀ����
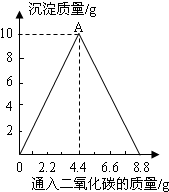
 25����֪��Ӧ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��ijʵ��С���ں�һ����Ca��OH��2�ij���ʯ��ˮ��ͨ��CO2������������������ͨ��CO2����
25����֪��Ӧ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��ijʵ��С���ں�һ����Ca��OH��2�ij���ʯ��ˮ��ͨ��CO2������������������ͨ��CO2�����������Ĺ�ϵ��ͼ��ʾ���ش��������⣺
��1��д��OA�η�Ӧ�Ļ�ѧ����ʽ��
Ca��OH��2+CO2�TCaCO3��+H2O
����2������ͨ��CO2��������Ϊ
8.8
gʱ����Һ������������ʵ���������Һ���������ֵ��������CO2������ˮ�е��ܽ⣩�������������⣬��Ϊ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��������ͼ��ʾ����ʼ������ɫ����������CO2�IJ���ͨ����������࣬Ȼ��������ϼ��٣������ȫ��ʧ��������д��Һ�ȱ���Ǻ��ֱ���壩
��1����ͼ��֪��OA�εij����������ӣ��ʿ�д��OA�η�Ӧ�Ļ�ѧ����ʽ��Ϊ������̼������������Һ�ķ�Ӧ��
��2������ʵ���������Һ���������ӣ��Ƕ�����̼���������ӣ��ڴ˹����У�����Ӧȫ���ܽ⣬���������ӲŻ���ܴ����ֵ��
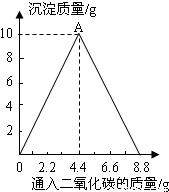
��1����ͼ��֪��OA�εij����������ӣ��ʿ�д��OA�η�Ӧ�Ļ�ѧ����ʽ��Ϊ������̼������������Һ�ķ�Ӧ��
��2������ʵ���������Һ���������ӣ��Ƕ�����̼���������ӣ��ڴ˹����У�����Ӧȫ���ܽ⣬���������ӲŻ���ܴ����ֵ��
����⣺�����⣬��Ϊ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��������ͼ��ʾ����Һ�ȱ���Ǻ��ֱ���壮������ǡ����Ƕ�����̼������������Һ��Ӧ����̼��Ƴ����Ĺ��̣������ֱ���塱��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2�Ĺ��̣�
��1����ͼ��֪��OA�εij����������ӣ��ʿ�д��OA�η�Ӧ�Ļ�ѧ����ʽΪCa��OH��2+CO2�TCaCO3��+H2O��
��2����Һ���������ӣ��Ƕ�����̼���������ӣ��ڴ˹����У�����Ӧȫ���ܽ⣬��Һ�����������ֵʱ����������ͨ��CO2��������Ϊ8.8��ʱ����Һ������������ʵ���������Һ���������ֵ��
�ʴ�Ϊ����1��Ca��OH��2+CO2�TCaCO3��+H2O��
��2��8.8�ˣ�
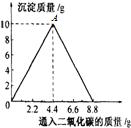
��1����ͼ��֪��OA�εij����������ӣ��ʿ�д��OA�η�Ӧ�Ļ�ѧ����ʽΪCa��OH��2+CO2�TCaCO3��+H2O��
��2����Һ���������ӣ��Ƕ�����̼���������ӣ��ڴ˹����У�����Ӧȫ���ܽ⣬��Һ�����������ֵʱ����������ͨ��CO2��������Ϊ8.8��ʱ����Һ������������ʵ���������Һ���������ֵ��
�ʴ�Ϊ����1��Ca��OH��2+CO2�TCaCO3��+H2O��
��2��8.8�ˣ�
���������ջ�ѧ����ʽ����д�����뷽�����˽ⳣ����ѧ��Ӧ�е�������ϵ��

��ϰ��ϵ�д�
 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
�����Ŀ
 ��֪��Ӧ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��ijʵ��С���ں�һ����Ca��OH��2�ij���ʯ��ˮ��ͨ��CO2������������������ͨ��CO2����
��֪��Ӧ��CaCO3+H2O+CO2=Ca��HCO3��2��������Ca��HCO3��2������ˮ��ijʵ��С���ں�һ����Ca��OH��2�ij���ʯ��ˮ��ͨ��CO2������������������ͨ��CO2����