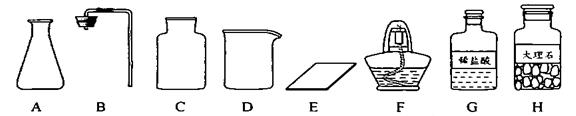
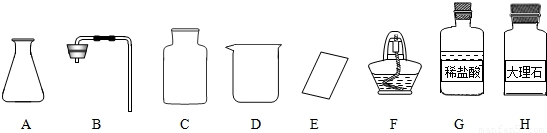
��Ŀ����
�γ���2013���п���ѧʵ�鿼���У�����������ȡ���ڶ�����̼����ȡ��������50 g��5����NaCl��Һ������������ǩ����ѧ����ǩȷ��һ��������п��飮
(1)��ͬѧ��ǩ������ʵ���ң����ֱ���ʵ��������Ҫ��������������ҩƷ��

����ͼ������D��������________��F��������________��
�ڼ�ͬѧ�鵽�Ŀ�ǩӦ����________(����ĸ����)��
A����������ȡ
B��������̼����ȡ
����ȡ������ķ�Ӧԭ��Ϊ________(�û�ѧ����ʽ��ʾ)����ȡһƿ�����壬Ӧѡ�õ�������________(����ĸ����)��
�ܼ�ͬѧʵ�����Ҫ����ʾ�����£���������������������װ�á���������ԡ�________���ռ����壮
�������������巢��װ�û�������ȡ�������壬��д������һ�ַ�Ӧ�Ļ�ѧ����
ʽ________��
(2)��ͬѧ����ɡ�����50 g��5����NaCl��Һ��ʵ������У�ͨ�����㣬�����NaCl________g����ȡˮԼ________mL���ܽ�NaClʱ�õ��IJ�������������________��
������
|
����(1)���ձ����ƾ��� ������B ������CaCO3��2HCl �������ȼӴ���ʯ���ϡ��� ������2H2O2 ����(2)2.5��47.5�����裬�ӿ��ܽ��ٶ� |

 �㾦�½̲�ȫ�ܽ��ϵ�д�
�㾦�½̲�ȫ�ܽ��ϵ�д� Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�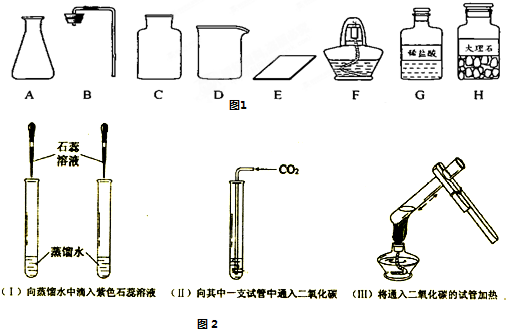
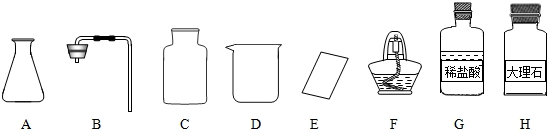
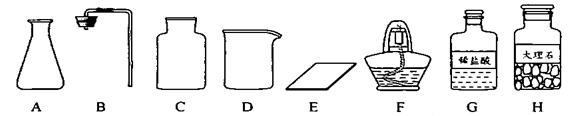 ��ͼ������D�������� ��F�������� ��
��ͼ������D�������� ��F�������� ��