��Ŀ����
ũ����Ա���á������䷽��������һƬ��������������⣬�����ʾ�ù���Ҫ����һ�����ĵ�Ԫ�غͼ�Ԫ�ء�
��1������ֲ����������ҪԪ�أ���������ȱ�ٵ�Ԫ�أ���Ҫ�õ��������䣬�������ڵ��ʵ��� (�����)��
��KCl�� ��NH4HCO3�� ��Ca3(PO4)2��
��2���������䷽������ʵ�֡�ȱʲô����ʲô����ʹ�øü����ĺô��� ��
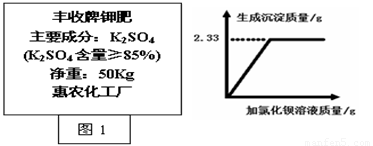
��3��ijͬѧȡ�����Ƽط�(��ͼ1)��Ʒ2��0g������������ˮ�У��μӹ����Ȼ�����Һ���������������������Ȼ�����Һ�����Ĺ�ϵ��ͼ2����ͨ�������жϸüط�������ص����������Ƿ���ϲ�Ʒ���װ˵��?(��Ӧ����ʽΪ��K2SO4+BaCl2��BaSO4��+2KCl�����ʲ��μӷ�Ӧ��)
��1���� ��2�֣� ��2����ֹ��Ⱦ����Լ��Դ ��2�֣�
��3��K2SO4 + BaCl2 �� BaSO4��+ 2KCl
174 233 174 ��233 = X :2��33 ��2�֣�
X 2��33�� X =1��74�� ��1�֣�
1��74��/2��0�� = 87% >85% ���� ��1�֣�
��������
�����������1��ֻ�Т��к��е�Ԫ�أ���ѡ�ڣ�2�������䷽����֪��������ȱ��ʲôԪ�أ�����������ԵĽ���ʩ�ʣ���Ч�ļ�����Ⱦ����Լ��Դ��3������2��33g���ᱵ���������������������ص�����Ϊ1��76g���ټ��������������Ϊ87%���Ӷ��ó�����
���㣺���ʵ����֡���ѧ����ʽ���������������ļ���


 ũ����Ա���á������䷽��������һƬ��������������⣬�����ʾ�ù���Ҫ����һ�����ĵ�Ԫ�غͼ�Ԫ�أ�
ũ����Ա���á������䷽��������һƬ��������������⣬�����ʾ�ù���Ҫ����һ�����ĵ�Ԫ�غͼ�Ԫ�أ�



