��Ŀ����
��������������Ҫ�Ŀ�ѧ������Ҳ�dz��õĻ�ѧѧϰ��������ѧ�Ͽ���ͨ�����ַ�Ӧ�õ�NH3(��������ˮ���ܶȱȿ���С)�����磺
��2NH4Cl(��)��Ca(OH)2(��)![]() CaCl2��2NH3����2H2O
CaCl2��2NH3����2H2O
��NH4HCO3(��)![]() NH3����CO2����H2O
NH3����CO2����H2O
��CaO(��)��NH3��H2O(Ũ)��NH3����Ca(OH)2
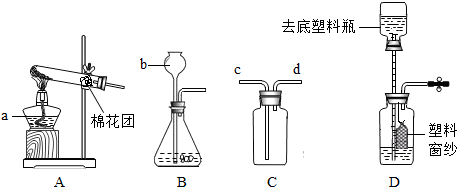
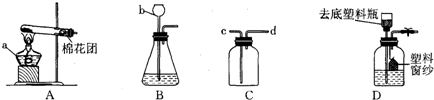
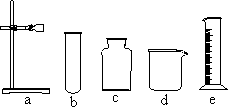
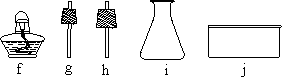
�ҽ�ѡ��Ӧ________(ֻдһ���������)��Ϊʵ������ȡNH3�ķ�Ӧԭ��������Ҫ�����������е�________(�����)��װ��NH3����װ�ã��Ҳ��õ��ռ�������________����ѡ�����ȡװ�û���������ȡ________���䷴Ӧ�Ļ�ѧ����ʽ��________��
�𰸣�
������
������
|
��(���),abfg(��abg��ch��hi),�����ſ�����,O2(��H2��CO2),2KClO3 |

��ϰ��ϵ�д�
�����Ŀ