��Ŀ����
�����ۺ�ͭ�۵ľ��Ȼ����ס��ҡ�����λͬѧ�ֱ�ȡ�û������ϡ���ᷴӦ�������������±���ʾ��| �� | �� | �� | |
| ȡ�ý�������������/g | 10 | 10 | 20 |
| ȡ��ϡ���������/g | 100 | 120 | 100 |
| ��Ӧ����ˣ��ø�����������/g | 4 | 4 | 13 |
��1���ҳ�ַ�Ӧ�����յõ���4g������______�����ѧʽ��
��2�������ͬѧ������Һ�����ʵ�������������д��������̣���������ȷ��0.1%��
��3����������Һ�к��е�������______�����ѧʽ��������______��
���𰸡���������1���Ƚϼ����������ݿ���֪��ͬ��10g�����ֱ���100gϡ���ᡢ120gϡ���ᷴӦʣ��������������4g�����Կ����ж�4g����Ϊͭ�����Ծݴ˽��
��2���Ƚϱ����е����ݿ���֪����10g�����к���ͭ������Ϊ4g����ô20g�����к���ͭ������Ϊ8g�������м���20g������100g���У�ʣ��Ĺ��������Ϊ13g�����Կ����жϽ���������е�����ʣ�࣬���Ծݴ˽�Ϸ�Ӧ�Ļ�ѧ����ʽ���м��㣻
��3���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4�����Ծݴ˽��
����⣺��1���Ƚϼ����������ݿ���֪��ͬ��10g�����ֱ���100gϡ���ᡢ120gϡ���ᷴӦʣ��������������4g�����Կ����ж�4g����Ϊͭ���仯ѧʽΪCu��
��2������������֪����Ӧ��������������Ϊ20g-13g=7g
��������������������Ϊx����������������Ϊy
Fe+H2SO4=FeSO4+H2��
56 152 2
7g x y
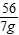 =
=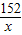 =
=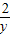
��ã�x=19g��y=0.25g
��Ӧ����Һ��������������������Ϊ��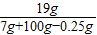 ×100%=17.8%��
×100%=17.8%��
�𣺱�ͬѧ������Һ��������������������Ϊ17.8%��
��3���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4�����Է�Ӧ����Һ������ΪH2SO4��FeSO4��
�ʴ�Ϊ����1��Cu��
��2��7.8%��
��3��H2SO4��FeSO4���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4��
������������Ҫ����ѧ��������ѧ��ѧ֪ʶ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������
��2���Ƚϱ����е����ݿ���֪����10g�����к���ͭ������Ϊ4g����ô20g�����к���ͭ������Ϊ8g�������м���20g������100g���У�ʣ��Ĺ��������Ϊ13g�����Կ����жϽ���������е�����ʣ�࣬���Ծݴ˽�Ϸ�Ӧ�Ļ�ѧ����ʽ���м��㣻
��3���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4�����Ծݴ˽��
����⣺��1���Ƚϼ����������ݿ���֪��ͬ��10g�����ֱ���100gϡ���ᡢ120gϡ���ᷴӦʣ��������������4g�����Կ����ж�4g����Ϊͭ���仯ѧʽΪCu��
��2������������֪����Ӧ��������������Ϊ20g-13g=7g
��������������������Ϊx����������������Ϊy
Fe+H2SO4=FeSO4+H2��
56 152 2
7g x y
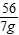 =
=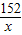 =
=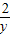
��ã�x=19g��y=0.25g
��Ӧ����Һ��������������������Ϊ��
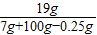 ×100%=17.8%��
×100%=17.8%���𣺱�ͬѧ������Һ��������������������Ϊ17.8%��
��3���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4�����Է�Ӧ����Һ������ΪH2SO4��FeSO4��
�ʴ�Ϊ����1��Cu��
��2��7.8%��
��3��H2SO4��FeSO4���ס����������ݶԱȿ�֪��������û����ȫ��Ӧ��������ϡ���ᷴӦ����FeSO4��
������������Ҫ����ѧ��������ѧ��ѧ֪ʶ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

��ϰ��ϵ�д�
 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д�
�����Ŀ
�����ۺ�ͭ�۵ľ��Ȼ����ס��ҡ�����λͬѧ�ֱ�ȡ�û������ϡ���ᷴӦ�������������±���ʾ��
| �� | �� | �� | |
| ȡ�ý�������������/g | 10 | 10 | 20 |
| ȡ��ϡ���������/g | 100 | 120 | 100 |
| ��Ӧ����ˣ��ø�����������/g | 4 | 4 | 13 |
��������ݣ��ش��������⣺
��1����������Һ�к������ʵ��� ��
��2������ʵ�����ݷ�������ȡ�ý��������15g�����뵽120gϡ�����У���ַ�Ӧ�����յõ���������Ϊ ��
��3�������ͬѧ������Һ�����ʵ�����������(д��������̣���������ȷ��0.1%��