��Ŀ����
ij��ѧ��ȤС��ͬѧ��ʵ�������ռ���һЩ����ΪFeSO4��CuSO4�ķ�Һ������л��ս���ͭ������������Һ����������·�����

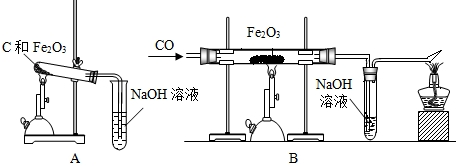
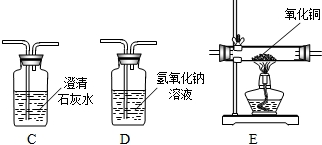
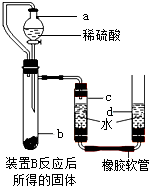
��1����������йط�Ӧ�Ļ�ѧ����ʽ�� _________ ��
��2��������������Լ�Y������Ϊ _________ ��
��3�������͢��ж��й��˲���������Ҫ�IJ��������� ________ �� _____ �� _______ ��
��4��������о������ˡ�ϴ�ӡ�������Եõ������Ľ���ͭ��ϴ��ʱ�������ͭ�Ƿ���ϴ���ķ����� _________ ��
��2��������������Լ�Y������Ϊ _________ ��
��3�������͢��ж��й��˲���������Ҫ�IJ��������� ________ �� _____ �� _______ ��
��4��������о������ˡ�ϴ�ӡ�������Եõ������Ľ���ͭ��ϴ��ʱ�������ͭ�Ƿ���ϴ���ķ����� _________ ��
��1��Fe+CuSO4=Cu+FeSO4
��2��ϡ����
��3��©�� �ձ� �������������Ⱥ�˳��
��4��ȡ���ϴ��Һ�������μ������Ȼ�����Һ�������ᱵ��Һ������������Һ������������������֤������ͭ��ϴ��
��2��ϡ����
��3��©�� �ձ� �������������Ⱥ�˳��
��4��ȡ���ϴ��Һ�������μ������Ȼ�����Һ�������ᱵ��Һ������������Һ������������������֤������ͭ��ϴ��

��ϰ��ϵ�д�
 �����Ļ�������ҵϵ�д�
�����Ļ�������ҵϵ�д�
�����Ŀ
 ��
�� ��������ͭ������㷺ʹ�õ����ֽ���������������ϢϢ��أ�ͼ1��ϡ��Ũ����ʵ���ʾ��ͼ��
��������ͭ������㷺ʹ�õ����ֽ���������������ϢϢ��أ�ͼ1��ϡ��Ũ����ʵ���ʾ��ͼ�� ��֪M��N�ֱ���ϡ���ᡢ����������Һ�е�һ�֣�ij��ѧ��ȤС��ͬѧ��һ������M�в��ϵμ�N�����ⶨ������Һ��pH����ͼ��ʾ��
��֪M��N�ֱ���ϡ���ᡢ����������Һ�е�һ�֣�ij��ѧ��ȤС��ͬѧ��һ������M�в��ϵμ�N�����ⶨ������Һ��pH����ͼ��ʾ��