��Ŀ����
2008�걱�����˻��������ڽ�����ʹ���˴����ĸ�����ij�������Ź�˾���е�1000t�����������������ù�˾�ú�Fe2O3 80%�ij�����ʯ������������������ش�
��1��д��������¯��һ����̼�ͳ�����Ӧ�Ļ�ѧ����ʽ��______
��2����������������������______��
��3���Ӹ�¯�����IJ�ƷΪ���������������Ƴɺ�̼������������͡��ߡ���______�ĸ֣�
��4��ͨ������˵���ù�˾����������������Ʒ��ó�������ٶ֣�������ʱ�����Ǹ����е������ɷ֣�
�⣺��1����������һ����̼�ڸ��������·�Ӧ���������Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ��Fe2O3+3CO 2Fe+3CO2 ��
2Fe+3CO2 ��
���Fe2O3+3CO 2Fe+3CO2 ��
2Fe+3CO2 ��
��2������������������������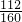 ��100%=70%��
��100%=70%��
�ó��������������������ǣ�80%��70%=56%��
���56%��
��3���ֵĺ�̼���������ĺ�̼���ͣ�
����ͣ�
��4��������������������Ʒ��ó����������ΪX��
X��56%=1000t��
X=1785.7t��
�𣺸ù�˾����������������Ʒ��ó�����1785.7�֣�
��������1�����ݷ�Ӧ������P�������غ㶨�ɿ�����д��ѧ����ʽ��
��2���������������������������������������Լ������������������������
��3�������еĺ�̼���ǣ�2-4.3%�����еĺ�̼���ǣ�0.03-2%��
��4�����ݸ����������������������������������Լ���������������
������������Ҫ���黯ѧ����ʽ����д����ط���ļ��㣬����ʱҪ���������ȷ���㣬����ó���ȷ�Ľ����
 2Fe+3CO2 ��
2Fe+3CO2 �����Fe2O3+3CO
 2Fe+3CO2 ��
2Fe+3CO2 ����2������������������������
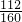 ��100%=70%��
��100%=70%���ó��������������������ǣ�80%��70%=56%��
���56%��
��3���ֵĺ�̼���������ĺ�̼���ͣ�
����ͣ�
��4��������������������Ʒ��ó����������ΪX��
X��56%=1000t��
X=1785.7t��
�𣺸ù�˾����������������Ʒ��ó�����1785.7�֣�
��������1�����ݷ�Ӧ������P�������غ㶨�ɿ�����д��ѧ����ʽ��
��2���������������������������������������Լ������������������������
��3�������еĺ�̼���ǣ�2-4.3%�����еĺ�̼���ǣ�0.03-2%��
��4�����ݸ����������������������������������Լ���������������
������������Ҫ���黯ѧ����ʽ����д����ط���ļ��㣬����ʱҪ���������ȷ���㣬����ó���ȷ�Ľ����

��ϰ��ϵ�д�
�����Ŀ


 29���ļ����˻���2008��8��8���ڱ���¡�ؾ��У������ҹ���һ��ʢ�£�
29���ļ����˻���2008��8��8���ڱ���¡�ؾ��У������ҹ���һ��ʢ�£�