��Ŀ����
����HCl��CaCl2�Ļ����Һ��Ϊ�˷��������Һ��HCl��CaCl2������������
���������ʵ�鷽����
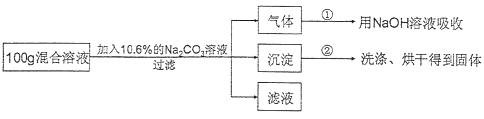
��ʵ�����ݡ�ʵ�鹲��¼���������ݣ��ڢ��飬����������ȫ��NaOH��Һ��������4.4g���ڢ��飬������ȫ�����ˡ�ϴ�ӡ���ɺ�õ����������Ϊ10g��
����ʵ����Ƽ��й����ݽ��з�������㣺
��1�������Һ��HCl����������Ϊ ����ֻд�����
��2�������Һ��CaCl2����������Ϊ���٣���д��������̣�
��3��ʵ���л����Һ��Na2CO3��Һǡ����ȫ��Ӧ�������˺����á���Һ�������ʵ���������Ϊ ���������ǹ����е���ʧ��ֻд���������С�����һλ��
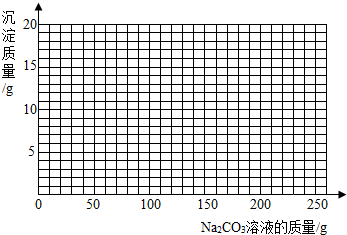
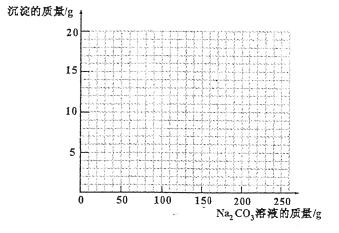
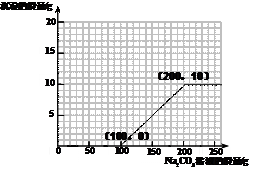
��4������100g�����Һ�в��ϼ���10.6% ��Na2CO3����Һ�����㻭������Na2CO3��Һ����������������������Ĺ�ϵͼ�����ڴ��������ͼ����ͼ��
��1��7.3%
��2���⣺������Һ��CaCl2������Ϊx��
CaCl2+Na2CO3��CaCO3��+2NaCl
111 100
x 10g
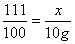
x = 11.1g
�����Һ��CaCl2����������=��11.1g��100g����100%=11.1%
�𣺻����Һ��CaCl2����������Ϊ11.1%��
��3��8.2%��1�֣�
|
��4����ͼ����ͼ���ߣ���1�֣�

 12�������£���Щ����CaCl2��NaCl�����ʵ�ˮ��Һ�����ԣ���Щ����Na2CO3�����ʵ�ˮ��Һ�ʼ��ԣ���Щ����NH4Cl�����ʵ�ˮ��Һ�����ԣ�����HCl��CaCl2�Ļ����Һ����������μ������ij����X����Һ��pH�����X�����ı仯��ϵ��ͼ��ʾ����X�ǣ�������
12�������£���Щ����CaCl2��NaCl�����ʵ�ˮ��Һ�����ԣ���Щ����Na2CO3�����ʵ�ˮ��Һ�ʼ��ԣ���Щ����NH4Cl�����ʵ�ˮ��Һ�����ԣ�����HCl��CaCl2�Ļ����Һ����������μ������ij����X����Һ��pH�����X�����ı仯��ϵ��ͼ��ʾ����X�ǣ�������