��Ŀ����
��5�֣��������ʾ��ͼ������������ʶ��ѧ���ʺ����⻯ѧ��Ӧ��ClO2����һ������ˮ�����������ҹ��ɹ����Ƴ��������ʹ������Ʒ�Ӧ��ȡClO2���·������䷴Ӧ���۹�����ͼ��ʾ��
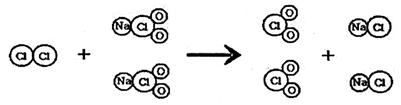
��1��д���÷�Ӧ�Ļ�ѧ����ʽ��__________________________________________��
��2���������������У�������������ǣ��ѧʽ��______________���Ȼ����������ӵķ��ſɱ�ʾΪ____________��
��3���ҹ��涨��������ˮ��ˮ�ʱ���ﵽ����ָ�꣺
a�����ó�����ɫ b����������ζ c��Ӧ������
���С�c��ָ�����ͨ�� ����������ƣ��ﵽ��
��1�� Cl2 + 2NaClO2 == 2NaCl + 2ClO2 ������ʽ2�֣�δ��ƽ��1�֣�
��2��ClO2 Na+
��3������
����������1����ͼ����Ϣ��֪�������ʹ������Ʒ�Ӧ���ɶ������Ⱥ��Ȼ��ƣ���Ӧ�Ļ�ѧ����ʽΪ��Cl2+2NaClO2�T2ClO2+2NaCl��
��2����ͼ����Ϣ��֪����������������Ԫ�غ���Ԫ����ɵĻ��������������Ȼ����е��������������ӣ�����Ϊ��Na+��
��3��ͨ�����˿ɽ�ˮ�в����ԵĹ������ʳ�ȥ���õ���������ˮ��

 ��ѧ�̸̳����¿α�ϵ�д�
��ѧ�̸̳����¿α�ϵ�д� Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д� ��ʾ��ԭ�ӣ���
��ʾ��ԭ�ӣ��� ��ʾ��ԭ�ӣ���
��ʾ��ԭ�ӣ��� ��ʾ
��ʾ
 ��ʾ
��ʾ
 ��ʾ
��ʾ


