��Ŀ����
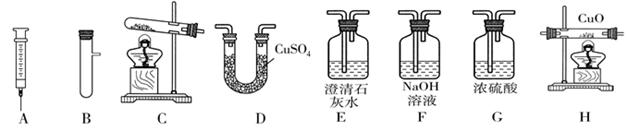
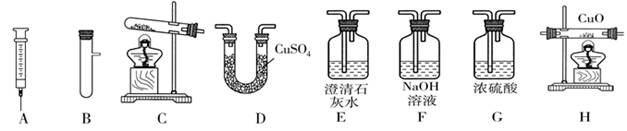
ij��ȤС��Ҫ�о�CO2������ЧӦ����ѡ�����к��ʵ�װ����գ�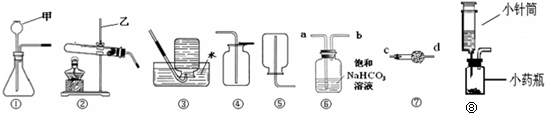
��1��д�����������Ƽ�______����______��
��2��ʵ�����ø��������ȡO2�Ļ�ѧ����ʽΪ______��Ӧѡ�ķ���װ����______������ţ�������ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ______��
��3��ʵ������ȡ���ռ������������CO2���������Ȼ����ˮ��������װ�õ�����˳����______��
��4��С���ü����С��Ͳ�ͷ�ҩƿ��װ��һ����װ�ã���ͼ��ʾ�����װ�â٣��ø�װ����ʵ����ŵ���______������ţ���
�ٽ�ԼҩƷ������������������ ������ȫ������Һ�ŷ�
���ܿ���Һ��ĵμ��ٶȡ����� �ܲ��������岻���κ����ʣ�
�⣺��1�����dz���©������������̨��
��2��ʵ�����ø��������ȡO2�Ļ�ѧ����ʽΪ2KMnO4 K2MnO4+MnO2+O2��������Ӧ���ڼ��ȹ�����ȡ���壬Ӧѡ�ķ���װ���Ǣڣ�����ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�����
K2MnO4+MnO2+O2��������Ӧ���ڼ��ȹ�����ȡ���壬Ӧѡ�ķ���װ���Ǣڣ�����ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�����
��3��Ҫ�õ���������Ķ�����̼Ӧ���ȳ����ٸ������ͨ�����͵�̼��������Һ�����Ȼ���������ͨ���߳�ȥˮ�������ܵ�����˳���ǡ������̳�����������˳���ǣ�abcd��
��4��װ�øĽ�����ŵ��ǣ�������㣻��ԼҩƷ�������ܿ���Һ��ĵμ��ٶȣ��Ӷ��ܹ����Ʒ�Ӧ�Ľ��У�
�ʴ�Ϊ����1������©��������̨����2��2KMnO4 K2MnO4+MnO2+O2�����ڣ��ų�����ƿ�еĿ�������3��abcd����4���٢ۣ�
K2MnO4+MnO2+O2�����ڣ��ų�����ƿ�еĿ�������3��abcd����4���٢ۣ�
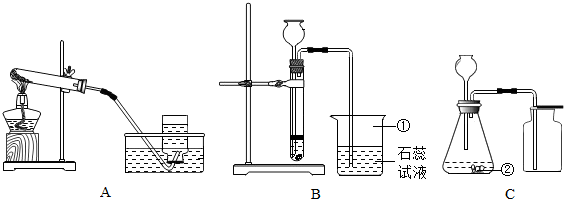
��������1������Ŀ�е�ͼ�Σ�����д���������������ƣ�
��2�����ݷ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã�����+���壬����Ҫ���ȣ�������д����Ӧԭ��������ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�����
��3��Ҫ�õ��������������Ӧ���ȳ����ٸ�����ų����ţ�
��4��ͼ��װ���ܿ���Һ��ĵμ��ٶȣ��Ӷ��ܹ����Ʒ�Ӧ�Ľ��У�
������������Ҫ�����������Ʒ����ռ������������������������ơ�װ�õĸĽ��ͻ�ѧ����ʽ����д����д��ѧ����ʽʱ��Ҫע����ƽ�������ʵ������˸Ľ�������ʵ������Դ�������ã����Ƚϻ�������Ҳ�ǽ�������п��ȵ�֮һ��
��2��ʵ�����ø��������ȡO2�Ļ�ѧ����ʽΪ2KMnO4
 K2MnO4+MnO2+O2��������Ӧ���ڼ��ȹ�����ȡ���壬Ӧѡ�ķ���װ���Ǣڣ�����ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�����
K2MnO4+MnO2+O2��������Ӧ���ڼ��ȹ�����ȡ���壬Ӧѡ�ķ���װ���Ǣڣ�����ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�������3��Ҫ�õ���������Ķ�����̼Ӧ���ȳ����ٸ������ͨ�����͵�̼��������Һ�����Ȼ���������ͨ���߳�ȥˮ�������ܵ�����˳���ǡ������̳�����������˳���ǣ�abcd��
��4��װ�øĽ�����ŵ��ǣ�������㣻��ԼҩƷ�������ܿ���Һ��ĵμ��ٶȣ��Ӷ��ܹ����Ʒ�Ӧ�Ľ��У�
�ʴ�Ϊ����1������©��������̨����2��2KMnO4
 K2MnO4+MnO2+O2�����ڣ��ų�����ƿ�еĿ�������3��abcd����4���٢ۣ�
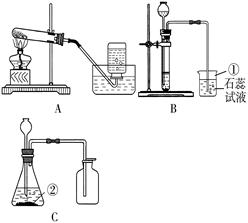
K2MnO4+MnO2+O2�����ڣ��ų�����ƿ�еĿ�������3��abcd����4���٢ۣ���������1������Ŀ�е�ͼ�Σ�����д���������������ƣ�
��2�����ݷ�Ӧ��״̬�ͷ�Ӧ����ѡ����װ�ã�����+���壬����Ҫ���ȣ�������д����Ӧԭ��������ˮ���ռ�����ʱ����ƿ��ˮ������Ϊ�ų�����ƿ�еĿ�����
��3��Ҫ�õ��������������Ӧ���ȳ����ٸ�����ų����ţ�
��4��ͼ��װ���ܿ���Һ��ĵμ��ٶȣ��Ӷ��ܹ����Ʒ�Ӧ�Ľ��У�
������������Ҫ�����������Ʒ����ռ������������������������ơ�װ�õĸĽ��ͻ�ѧ����ʽ����д����д��ѧ����ʽʱ��Ҫע����ƽ�������ʵ������˸Ľ�������ʵ������Դ�������ã����Ƚϻ�������Ҳ�ǽ�������п��ȵ�֮һ��

��ϰ��ϵ�д�
�����Ŀ
ͬѧ��ѧϰ�ˡ���������һ�ºԽ������й����ʲ�����Ũ�����Ȥ��
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
��2�����Ӳ�ʲ������ں�ɫ��ĩΪ�������ʵĻ���Ҫȷ����ĩ���������ʵ���������������Ҫͨ���й����ݼ�������жϣ���������Ϊ�������ݵ��ǣ� ��
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a b�������������=����
�ڲ������������ʣ�a b�������������=����
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺
����Ʒ��ͭ����������Ϊ�� ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ �ˣ�
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
| ���� | ��ʵ�鷽�� | ���� | CO��CuO��Ӧ�Ļ�ѧ����ʽ |
| ��ĩΪCu | ����Ӧ | CuO+CO=Cu+CO2 | |
| ��ĩ�ܽ⣬��Һ�����ɫ�����ɺ�ɫ��ĩ | 2CuO+CO=Cu2O+CO2 |
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a
�ڲ������������ʣ�a
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺

����Ʒ��ͭ����������Ϊ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ
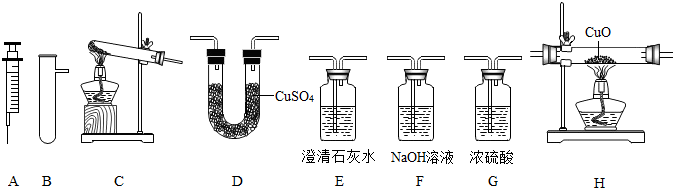
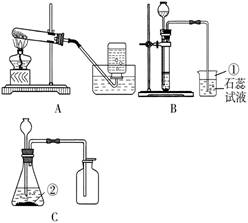
 ij��ȤС����������װ�ý���O2��CO2��ʵ�����Ʒ����й����ʵ��о���
ij��ȤС����������װ�ý���O2��CO2��ʵ�����Ʒ����й����ʵ��о���