��Ŀ����
��һ��ɽ�ϵ�ʯ��ʯ��Ʒ��ֻ�����ʶ������裨����������һ�ּȲ�����ˮ��Ҳ�������ᷴӦ�Ĺ��壩��С�պ�����ͬѧ��ⶨ����Ʒ��̼��Ƶ���������������ȡһ��ʯ��ʯ��Ʒ���������Ƴ�9��4g�����ձ��ڣ��ձ�������Ϊ60��8g����Ȼ����������ϡ���ᣬ�ò��������������ٲ�������Ϊֹ������ȥϡ����97��8g���ٳƳ���Ӧ���ձ����������ʵ�������Ϊ164��7g���Իش�
��1����ʯ��ʯ��Ʒ�������ҪĿ����????????????????????????????????????? ��
��2��ʵ��������ų�������̼???????? g�������ܽ���ˮ�еĶ�����̼�������Բ��ƣ���
��3����ʯ��ʯ��Ʒ��̼��Ƶ����������Ƕ��٣�����������ȷ��0��1%��
��4����Ӧ����Һ������Ϊ??????????? g��
��1��Ϊ������Ӧ��ĽӴ������ʹ��Ӧ�ܿ��ٽ���
��2��3��3g
��3��79��8%
��4��102
��������
�����������1��Ϊ������Ʒ��̼��Ʋ�����ɷ�Ӧ���ɰ���Ʒ���飬������Ʒ������ĽӴ�������˾ٻ����Լӿ췴Ӧ�����ʣ�
��2�����������غ㶨�ɿ���֪���ų����������̼������=9��4g+60��8g+97��8g-164��7g=3��3g
��3����ʯ��ʯ��Ʒ��̼��Ƶ�����Ϊx
CaCO3+2HCl�TCaCl2+H2O+CO2��
100?????????????????? ��44
x???????????????????? ��2��2g
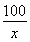 =
= 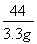
��ã�x=7��5g????
��ʯ��ʯ��w(CaCO3)= 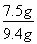 ��100%=79��8%
��100%=79��8%
�𣺸�ʯ��ʯ��Ʒ��̼��Ƶ�����������79��8%��
��4����Ӧ����Һ������Ϊ��164��7g-60��8g-��9��4g-7��5g��=102g
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���

 ��У����ϵ�д�
��У����ϵ�д� ��2005?ɽ������ʡ���ض��зḻ��ʯ��ʯ�����Դ����һ��ɽ�ϵ�ʯ��ʯ��Ʒ������ֻ�����ʶ������裨����������һ�ּȲ�����ˮҲ�������ᷴӦ�����µĹ��壩��С�պ�����ͬѧ��ⶨ����Ʒ��̼��Ƶ���������������ȡһ��ʯ��ʯ��Ʒ���������Ƴ�6g�����ձ��ڣ��ձ�������Ϊ20g����Ȼ�����50g����ϡ���ᣬ�ò��������������ٲ�������Ϊֹ����Ӧ����ʱ�䣨t�����ձ�������ʢ���ʵ���������m���Ĺ�ϵ��ͼ��ʾ��
��2005?ɽ������ʡ���ض��зḻ��ʯ��ʯ�����Դ����һ��ɽ�ϵ�ʯ��ʯ��Ʒ������ֻ�����ʶ������裨����������һ�ּȲ�����ˮҲ�������ᷴӦ�����µĹ��壩��С�պ�����ͬѧ��ⶨ����Ʒ��̼��Ƶ���������������ȡһ��ʯ��ʯ��Ʒ���������Ƴ�6g�����ձ��ڣ��ձ�������Ϊ20g����Ȼ�����50g����ϡ���ᣬ�ò��������������ٲ�������Ϊֹ����Ӧ����ʱ�䣨t�����ձ�������ʢ���ʵ���������m���Ĺ�ϵ��ͼ��ʾ��