��Ŀ����
���������Ǵ�����Ⱦ��֮һ���ҹ��Ļ��������������жԿ����ж�����������Ũ�ȣ���λ����Ŀ��������������������������ֵ���ұ���ʾ��

Ϊ�ⶨij�ؿ����ж�������ĺ�����ij��ѧ����С���������ʵ�飺���Թ��м���һ�����ĺ��⣨I2��1.27mg�ĵ���Һ���ټ���2~3�ε�����Һ��������I2����ɫ�������Թ���ͨ�����������Һ����ɫ��Ϊ��ɫʱǡ����ȫ��Ӧ������ȥ�������Ϊ1000L����ͨ�������жϳ��˿����ж��������Ũ�ȼ��𡣣���ѧ����ʽΪ��SO2+I2+2H2O=H2SO4+2HI��
���𰸡�
��5�֣�ͨ������SO2��Ũ��Ϊ0.32mg/m3 ��Ϊ������
����������

��ϰ��ϵ�д�
 ������ϰ�ο����뵥Ԫ���ϵ�д�
������ϰ�ο����뵥Ԫ���ϵ�д�
�����Ŀ
 ���������Ǵ�����Ⱦ��֮һ���ҹ��Ļ��������������жԿ����ж�����������Ũ�ȣ���λ����Ŀ��������������������������ֵ�����ʾ��
���������Ǵ�����Ⱦ��֮һ���ҹ��Ļ��������������жԿ����ж�����������Ũ�ȣ���λ����Ŀ��������������������������ֵ�����ʾ��| Ũ����ֵ��mg/m3�� |
| һ���� ������ ������ 0.15 0.50 0.70 |
 ���������Ǵ�����Ⱦ��֮һ���ҹ��Ŀ����������жԿ����ж�����������Ũ�ȣ���λ����Ŀ��������������������������ֵ���±���ʾ��
���������Ǵ�����Ⱦ��֮һ���ҹ��Ŀ����������жԿ����ж�����������Ũ�ȣ���λ����Ŀ��������������������������ֵ���±���ʾ��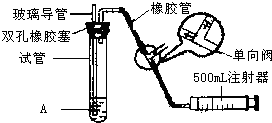 ���������Ǵ�����Ⱦ��֮һ��Ϊ���Եزⶨ��Χ�����е�SO2������ijѧ������С���������ͼ��ʵ��װ�ã�
���������Ǵ�����Ⱦ��֮һ��Ϊ���Եزⶨ��Χ�����е�SO2������ijѧ������С���������ͼ��ʵ��װ�ã�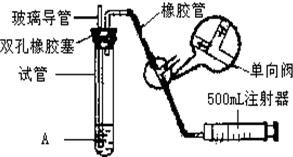 ���������Ǵ�����Ⱦ��֮һ��Ϊ���Եزⶨ��Χ�����е�SO2������ijѧ������С���������ͼ��ʵ��װ�ã�
���������Ǵ�����Ⱦ��֮һ��Ϊ���Եزⶨ��Χ�����е�SO2������ijѧ������С���������ͼ��ʵ��װ�ã�