��Ŀ����
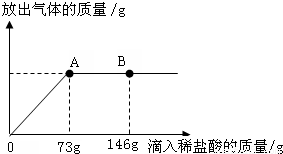
��һ�ձ���ʢ��22.3Na2CO3��NaCl��ɵĹ����������109.1g ˮʹ����ȫ�ܽ⣬�����Һ���������μ�������������Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺��1�����μ���73gϡ����ʱ���ų������������Ϊ______g��
��2�����μ�ϡ������ͼ��B��ʱ���ձ�����Һ��������ǣ�д��ѧʽ��______��
��3�����μ���73gϡ����ʱ����A��ʱ�����ձ���Ϊ��������Һ����ͨ���������������Һ�����ʵ�����������
���𰸡���������1����ΪNaCl������ܻ��ཻ���ɷ֣����Բ���Ӧ�����ֻҪ�����̼������73gϡ���ᷴӦ���ɵ������������ɣ����ݷ�Ӧ�Ļ�ѧ����ʽ���ó�������֮��������ȣ��г�����ʽ���Ϳɼ�����ų������������
��2����ͼʾ��֪�����μ�ϡ������ͼ��A��ʱ��������ȫ��Ӧ���ٵμ�ϡ������ͼ��B��ʱ���������ʣ�࣬��ʱ��Һ�е����ʾ����Ȼ��ƺ��Ȼ��⣮
��3���������������A��ʱ������ȫ��Ӧ����Һ��������л������ԭ�е��Ȼ��ƺ��������е��Ȼ��ƣ����ݷ�Ӧ�Ļ�ѧ����ʽ���ó�������֮��������ȣ��г�����ʽ���Ϳɼ�����������Na2CO3�������ͷ�Ӧ���ɵ�NaCl����������������������ȥNa2CO3���������ǻ������ԭ�е��Ȼ��Ƶ��������ټ��Ϸ�Ӧ���ɵ�NaCl�����������������ʵ�������
����⣻��1����ų������������Ϊz��
Na2CO3+2HCl�T2NaCl+H2O+CO2��
73 44
73g×10% z

��֮�ã�z=4.4g
��2����ͼʾ��֪�����μ�ϡ������ͼ��A��ʱ��������ȫ��Ӧ���ٵμ�ϡ������ͼ��B��ʱ���������ʣ�࣮����Һ�е�����ΪNaCl��HCl��
��3���⣺�跴Ӧ������NaCl������Ϊx���������Na2CO3������Ϊy��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 73 117
y 73g×10% x

��֮�ã�y=10.6g x=11.7g
��Ӧ������NaCl������=11.7g+22.3g-10.6g=23.4g����Һ��������Ϊ22.3g+109.1g+73g-4.4g=200g����������Һ�����ʵ���������Ϊ ×100%=11.7%
×100%=11.7%
�ʴ�Ϊ����1��4.4����2��NaCl��HCl����3��������Һ�����ʵ���������Ϊ11.7%��
����������ϺõĿ���ѧ������ͼ����������ѧ��Ӧ��������ѧ��Ӧ��ȷͼ���еĹؼ��㼰�ߵı仯����ʾ�ĺ��壬��ͼ��ͻ�ѧ��Ӧ���ܽ���ǽ���Ĺؼ����ڣ�
��2����ͼʾ��֪�����μ�ϡ������ͼ��A��ʱ��������ȫ��Ӧ���ٵμ�ϡ������ͼ��B��ʱ���������ʣ�࣬��ʱ��Һ�е����ʾ����Ȼ��ƺ��Ȼ��⣮
��3���������������A��ʱ������ȫ��Ӧ����Һ��������л������ԭ�е��Ȼ��ƺ��������е��Ȼ��ƣ����ݷ�Ӧ�Ļ�ѧ����ʽ���ó�������֮��������ȣ��г�����ʽ���Ϳɼ�����������Na2CO3�������ͷ�Ӧ���ɵ�NaCl����������������������ȥNa2CO3���������ǻ������ԭ�е��Ȼ��Ƶ��������ټ��Ϸ�Ӧ���ɵ�NaCl�����������������ʵ�������
����⣻��1����ų������������Ϊz��
Na2CO3+2HCl�T2NaCl+H2O+CO2��
73 44
73g×10% z

��֮�ã�z=4.4g
��2����ͼʾ��֪�����μ�ϡ������ͼ��A��ʱ��������ȫ��Ӧ���ٵμ�ϡ������ͼ��B��ʱ���������ʣ�࣮����Һ�е�����ΪNaCl��HCl��
��3���⣺�跴Ӧ������NaCl������Ϊx���������Na2CO3������Ϊy��
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 73 117
y 73g×10% x

��֮�ã�y=10.6g x=11.7g
��Ӧ������NaCl������=11.7g+22.3g-10.6g=23.4g����Һ��������Ϊ22.3g+109.1g+73g-4.4g=200g����������Һ�����ʵ���������Ϊ
 ×100%=11.7%
×100%=11.7%�ʴ�Ϊ����1��4.4����2��NaCl��HCl����3��������Һ�����ʵ���������Ϊ11.7%��
����������ϺõĿ���ѧ������ͼ����������ѧ��Ӧ��������ѧ��Ӧ��ȷͼ���еĹؼ��㼰�ߵı仯����ʾ�ĺ��壬��ͼ��ͻ�ѧ��Ӧ���ܽ���ǽ���Ĺؼ����ڣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��һ�ձ���ʢ��22.3gNa2CO3��NaCl��ɵĹ�������������μ�������������Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺
��һ�ձ���ʢ��22.3gNa2CO3��NaCl��ɵĹ�������������μ�������������Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺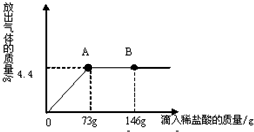
 ��һ�ձ���ʢ��22.3g Na2CO3��NaCl��ɵĹ��������143.1gˮ�ܽ⣬�Ƴ���Һ��
��һ�ձ���ʢ��22.3g Na2CO3��NaCl��ɵĹ��������143.1gˮ�ܽ⣬�Ƴ���Һ�� ��2013?��������һģ����һ�ձ���ʢ��22.3Na2CO3��NaCl��ɵĹ����������109.1g ˮʹ����ȫ�ܽ⣬�����Һ���������μ�������������Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺
��2013?��������һģ����һ�ձ���ʢ��22.3Na2CO3��NaCl��ɵĹ����������109.1g ˮʹ����ȫ�ܽ⣬�����Һ���������μ�������������Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ������ͼ��ʾ�����������ش����⣺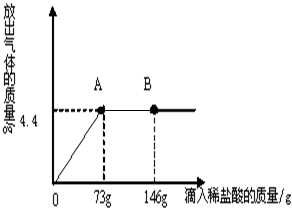 С���ڻ�ѧʵ���ҷ��֣�ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ��С�ս���С����С�죬��ͬ̽�����ְ�ɫ��ĩ�ijɷ֣�
С���ڻ�ѧʵ���ҷ��֣�ʢ��NaOH��Һ���Լ�ƿƿ�ں���Ƥ���ϳ����˰�ɫ��ĩ��С�ս���С����С�죬��ͬ̽�����ְ�ɫ��ĩ�ijɷ֣�