��Ŀ����
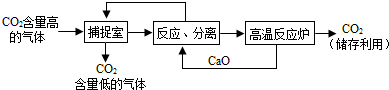
�����п�ѧ��������á�̼��������������ҵ�����ж�����̼���ŷ�������̼����������ָͨ��һ���ķ���������ҵ�����в�����CO2����������д�������ã�������������NaOH��Һ��������CO2����������ͼ��ʾ����������������δ�������

�ٲ����з�����Ӧ�Ļ�ѧ����ʽΪ________��
�ڴ˷�Ӧ���������׳�________������������ˮ�Ĺ��̻�________�����Ȼ����ȣ�
�ۡ���Ӧ���롱�У��õ��������ʵĻ���������________���ù�����̼��ƣ�
�����������У�����ѭ�����õ�������________��
CO2+2NaOH=Na2CO3+H2O ��ʯ�� ���� ���� �������ƺ�������
���������ݶ�����̼���������Ʒ�Ӧ�ķ�Ӧ�������д����ѧ����ʽ����ʯ�ҿ���ˮ��Ӧ�����ų��������ȣ������ǽ������Һ�����ķ�����������ͼ������ѭ�����õ����ʣ�
��𣺢ٲ����з�����Ӧ�Ļ�ѧ��Ӧ�Ƕ�����̼������������Һ��Ӧ����̼���ƺ�ˮ����ѧ����ʽΪCO2+2NaOH=Na2CO3+H2O��
���CO2+2NaOH=Na2CO3+H2O��
���������׳���ʯ�ң�CaO��ˮ��Ӧ�����������Ʋ��ų��������ȣ�
�����ʯ�ң����ȣ�
�۹�����Һ��ֿ��ù��ˣ�
������ˣ�
��̼�������������Ʒ�Ӧ����̼��Ƴ������������ƣ������������Dz����еķ�Ӧ����Կ���ѭ�����ã�̼��Ƹ������������ƺͶ�����̼���۲�ͼʾ�������ƻ���ѭ�����ã������ƺ��������ƶ�����ѭ�����ã�
����������ƺ������ƣ�
�����������Ƕ����ʵ����ʺ�����ͼ�ķ�����ע�ض�ѧ������������Ŀ��飬����������ѧ����̽��������
���������ݶ�����̼���������Ʒ�Ӧ�ķ�Ӧ�������д����ѧ����ʽ����ʯ�ҿ���ˮ��Ӧ�����ų��������ȣ������ǽ������Һ�����ķ�����������ͼ������ѭ�����õ����ʣ�
��𣺢ٲ����з�����Ӧ�Ļ�ѧ��Ӧ�Ƕ�����̼������������Һ��Ӧ����̼���ƺ�ˮ����ѧ����ʽΪCO2+2NaOH=Na2CO3+H2O��
���CO2+2NaOH=Na2CO3+H2O��
���������׳���ʯ�ң�CaO��ˮ��Ӧ�����������Ʋ��ų��������ȣ�
�����ʯ�ң����ȣ�
�۹�����Һ��ֿ��ù��ˣ�
������ˣ�
��̼�������������Ʒ�Ӧ����̼��Ƴ������������ƣ������������Dz����еķ�Ӧ����Կ���ѭ�����ã�̼��Ƹ������������ƺͶ�����̼���۲�ͼʾ�������ƻ���ѭ�����ã������ƺ��������ƶ�����ѭ�����ã�
����������ƺ������ƣ�
�����������Ƕ����ʵ����ʺ�����ͼ�ķ�����ע�ض�ѧ������������Ŀ��飬����������ѧ����̽��������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ