��Ŀ����
�±���Ԫ�����ڱ���һ���֣�����ϸ�Ķ�����գ�
| �� ���� | �� | 0 | ||||||
| 1 | 1 H 1.008 | �� | �� | �� | �� | �� | �� | 2 He 4.003 |
| 2 | 3 Li 6.942 | 4 Be 9.012 | 5 B 10.81 | 6 C 12.02 | 7 N 14.01 | 8 O 16.00 | 9 F 19.00 | 10 Ne 20.15 |
| 3 | 11 Na 22.99 | 12 Mg 24.31 | 13 | 14 Si 28.09 | 15 P 30.97 | 16 S 32.06 | 17 Cl 35.5 | 18 Ar 39.95 |
��2����Ԫ���ڵ�______���ڣ��ڻ�ѧ��Ӧ����______���ӣ���õ�����ʧȥ������
��3����Ԫ������е�______Ԫ�صĻ�ѧ�������ƣ�
��4������ԭ������Ϊ13��Ԫ����______Ԫ�أ�
�⣺��1������Ԫ�����ڱ��ɲ��þԪ�ص����ԭ������Ϊ��24.31�� �ʴ�Ϊ��24.31��
��2��ͨ��Ԫ�����ڱ��в��ң�Ԫ�����ڱ��к���ָ���ڣ�����ָ�����壬�������������жϵ�ʧ����������ʴ�Ϊ�������õ���
��3������Ԫ�������ɣ�ͬһ���У�ԭ��������������ͬ��������������ͬ��Ԫ�ػ�ѧ�������ƣ��ɴ˿ɵã����Ԫ�ػ�ѧ�������Ƶ�Ԫ������Ԫ�أ��ʴ�Ϊ���Ȼ�Cl��
��4������ԭ������Ϊ13��Ԫ��������AlԪ�أ��ʴ�Ϊ������Al��
��������1������Ԫ�����ڱ����ɽ���⣻
��2��ͨ��Ԫ�����ڱ��в��ң�Ԫ�����ڱ��к���ָ���ڣ�����ָ�����壬�������������жϵ�ʧ���������
��3������������������ͬ��Ԫ�ػ�ѧ�������ƽ��н���⣮
��4��ͨ��Ԫ�����ڱ��в��ң�ԭ������Ϊ13��Ԫ�أ�
���������⿼��ѧ����������Ӧ��Ԫ�����ڱ����ṩ����Ϣ���з����������������Ϣ�����������Ź�����Ϣ����ȡ��������ֱ�ӽ�����б�Ҫ��������Ŀ��Ӧ�Բ���Ӧ���Ķ�---����---�ۺ�������ԭ�����·����������
��2��ͨ��Ԫ�����ڱ��в��ң�Ԫ�����ڱ��к���ָ���ڣ�����ָ�����壬�������������жϵ�ʧ����������ʴ�Ϊ�������õ���
��3������Ԫ�������ɣ�ͬһ���У�ԭ��������������ͬ��������������ͬ��Ԫ�ػ�ѧ�������ƣ��ɴ˿ɵã����Ԫ�ػ�ѧ�������Ƶ�Ԫ������Ԫ�أ��ʴ�Ϊ���Ȼ�Cl��
��4������ԭ������Ϊ13��Ԫ��������AlԪ�أ��ʴ�Ϊ������Al��
��������1������Ԫ�����ڱ����ɽ���⣻
��2��ͨ��Ԫ�����ڱ��в��ң�Ԫ�����ڱ��к���ָ���ڣ�����ָ�����壬�������������жϵ�ʧ���������
��3������������������ͬ��Ԫ�ػ�ѧ�������ƽ��н���⣮
��4��ͨ��Ԫ�����ڱ��в��ң�ԭ������Ϊ13��Ԫ�أ�
���������⿼��ѧ����������Ӧ��Ԫ�����ڱ����ṩ����Ϣ���з����������������Ϣ�����������Ź�����Ϣ����ȡ��������ֱ�ӽ�����б�Ҫ��������Ŀ��Ӧ�Բ���Ӧ���Ķ�---����---�ۺ�������ԭ�����·����������

��ϰ��ϵ�д�
�����Ŀ
�±���Ԫ�����ڱ���һ���֣�
��1��16��Ԫ�ص�Ԫ�ط���Ϊ ����Ԫ�ص�ԭ�ӽṹʾ��ͼ���ң���X����ֵ= ��
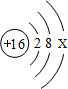
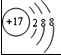
��2��8��Ԫ�غ�13��Ԫ����ɵĻ�����Ļ�ѧʽΪ ��
��3�����ñ��е�Ԫ�ط�������ɵķ�Ӧ��д������Ҫ��Ļ�ѧ����ʽ����ˮ���ɵĻ��Ϸ�Ӧ�� ��
| �� ���� |
IA | 0 | ||||||
| һ | 1H 1��008 |
��A | ��A | ��A | V A | ��A | ��A | 2He 4��003 |
| �� | 3Li 6��941 |
4Be 9��012 |
5B 10��81 |
6C 12��01 |
7N 14��01 |
8O 16��00 |
9F 19��00 |
10Ne 20��18 |
| �� | 11Na 22��99 |
12Mg 24��31 |
13Al 26��98 |
14Si 28��09 |
15P 30��97 |
16S 32��06 |
17Cl 35��45 |
18Ar 39��95 |
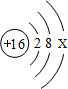
��2��8��Ԫ�غ�13��Ԫ����ɵĻ�����Ļ�ѧʽΪ
��3�����ñ��е�Ԫ�ط�������ɵķ�Ӧ��д������Ҫ��Ļ�ѧ����ʽ����ˮ���ɵĻ��Ϸ�Ӧ��


 ����m=
����m=
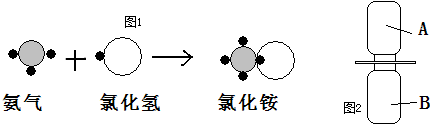
 ��ʾ����
��ʾ���� ����ʾ��ԭ�ӣ��á�
����ʾ��ԭ�ӣ��á� ����ʾ��ԭ�ӣ��á�
����ʾ��ԭ�ӣ��á� ����ʾ��ԭ�ӣ�������Ӧ���̿���ͼ1��ʾ��
����ʾ��ԭ�ӣ�������Ӧ���̿���ͼ1��ʾ��