��Ŀ����
������һ����Ҫ�IJ��ϣ������������������벻����������ͼ��ʾ�˽������˳�����ͭ������������Ԫ�ر�������ģ���������õĴ������ޣ�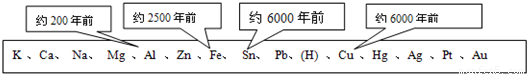
��1������ͼ�����ݺ��йصĻ�ѧ֪ʶ������Ϊ�������ģ���������õ��Ⱥ�˳���������Щ�����й�______��
�ٵؿ��н���Ԫ�صĺ��� �ڽ����ĵ����� �۽����Ļ�� �ܽ�������չ�� �ݽ���ұ�������׳̶�
��2�����Ļ�Ա���ǿ�������ڿ�����ȴ���ֳ����õĿ�ʴ�ԣ���ԭ����ʲô��
��3��С����ⶨCu-Zn�Ͻ��Cu-Ag�Ͻ���ͭ������������ʵ����ֻ�ṩ��һƿϡ����ͱ�Ҫ��������
������Ϊ���ܲ��ͭ�����������ĺϽ���______�Ͻ�
��С��ȡ�úϽ�ķ�ĩ32.5g���������������ַ�Ӧ���ⶨ������0.4g���壬���������������úϽ���ͭ������������
���𰸡���������1���������еĽ�����֪ʶ���з����������Ļ��Խǿ��ұ���ij̶�Խ�ѣ����õ�Խ����
��2�����Ļ�Ա���ǿ����������ʴ��Ҫ���γɱ���Ĥ�Ƕȿ��ǣ�
��3�������ڽ������˳���У���ǰ�Ľ��������ᷢ���û���Ӧ����������������������������������������úϽ���п���������Ӷ����ͭ��������ͭ������������
����⣺��1�������֪�ؿ��ﺬ����ߵ��ǽ������������������õ�ʱ���ں�˵���������ģ���������õ��Ⱥ�˳����ؿ�����������أ�ͬʱ������ĵ����Ժ���չ��Ҳ�أ�������������˳��ͽ���ұ�������׳̶��йأ������Ļ��Խǿ��ұ���ij̶�Խ�ѣ����õ�Խ������ѡ�ۢݣ�
��2�����Ļ�Ա���ǿ�������ڿ�����ȴ���ֳ����õĿ���ʴ�ԣ�����Ϊ������������е�����������Ӧ��������һ�����ܵ�������Ĥ���������ı��棬��ֹ��Ӧ�Ľ��У����Ծ������õĿ���ʴ�ԣ�
�ʴ�Ϊ��������������е�����������Ӧ��������һ�����ܵ�������Ĥ���������ı��棬��ֹ��Ӧ�Ľ��У����Ծ������õĿ���ʴ�ԣ�
��3����ֻ��������ͱ�Ҫ����������Ͻ����躬���������ᷴӦ�Ľ�����п�������ǰ�棬��ͭ����������ĺ��棬�������ᷴӦ������ֻ�ܲ��ͭп�Ͻ�
�ʱ����Ϊ��Cu��Zn��
��������0.4g������Ҫп������Ϊx������
Zn+2HCl�TZnCl2+H2��
65 2
x 0.4g
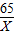 =
=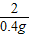
��֮�ã�x=13g
�úϽ��е�Cu������������=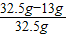 ×100%=60%��
×100%=60%��
�ʸúϽ���ͭ����������Ϊ60%��
����������ӽ��������ã���ʴ�ȷ���ȫ�濼���˽��������ʣ����⿼���˽����˳��������Լ����ݻ�ѧ����ʽ���еļ��㣬�ڽ�ڶ���ʱҪע�����Ĺ淶�ԣ�
��2�����Ļ�Ա���ǿ����������ʴ��Ҫ���γɱ���Ĥ�Ƕȿ��ǣ�
��3�������ڽ������˳���У���ǰ�Ľ��������ᷢ���û���Ӧ����������������������������������������úϽ���п���������Ӷ����ͭ��������ͭ������������
����⣺��1�������֪�ؿ��ﺬ����ߵ��ǽ������������������õ�ʱ���ں�˵���������ģ���������õ��Ⱥ�˳����ؿ�����������أ�ͬʱ������ĵ����Ժ���չ��Ҳ�أ�������������˳��ͽ���ұ�������׳̶��йأ������Ļ��Խǿ��ұ���ij̶�Խ�ѣ����õ�Խ������ѡ�ۢݣ�
��2�����Ļ�Ա���ǿ�������ڿ�����ȴ���ֳ����õĿ���ʴ�ԣ�����Ϊ������������е�����������Ӧ��������һ�����ܵ�������Ĥ���������ı��棬��ֹ��Ӧ�Ľ��У����Ծ������õĿ���ʴ�ԣ�
�ʴ�Ϊ��������������е�����������Ӧ��������һ�����ܵ�������Ĥ���������ı��棬��ֹ��Ӧ�Ľ��У����Ծ������õĿ���ʴ�ԣ�
��3����ֻ��������ͱ�Ҫ����������Ͻ����躬���������ᷴӦ�Ľ�����п�������ǰ�棬��ͭ����������ĺ��棬�������ᷴӦ������ֻ�ܲ��ͭп�Ͻ�
�ʱ����Ϊ��Cu��Zn��
��������0.4g������Ҫп������Ϊx������
Zn+2HCl�TZnCl2+H2��
65 2
x 0.4g
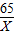 =
=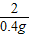
��֮�ã�x=13g
�úϽ��е�Cu������������=
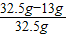 ×100%=60%��
×100%=60%���ʸúϽ���ͭ����������Ϊ60%��
����������ӽ��������ã���ʴ�ȷ���ȫ�濼���˽��������ʣ����⿼���˽����˳��������Լ����ݻ�ѧ����ʽ���еļ��㣬�ڽ�ڶ���ʱҪע�����Ĺ淶�ԣ�

��ϰ��ϵ�д�
 ������ÿ�ʱ��ҵϵ�д�
������ÿ�ʱ��ҵϵ�д�
�����Ŀ




 ������һ����Ҫ�IJ��ϣ������������������벻��������
������һ����Ҫ�IJ��ϣ������������������벻��������