��Ŀ����
2011��11��28����12��9�գ������Ϲ�����仯��ܹ�Լ����17�ε�Լ���������ϷǶ����������е°���У��й��������ڼ��ŵij�ŵ�����չʾ���й�ı��չ���ٺ����������εĴ������Ŀǰ������̼������Ϊ����Ĺ�ʶ����1�������һ�����ճ������з��ϡ���̼�����ʽ��������______��
��2����ѧ�Ҵ�ȡ�����ת������������CO2��H2��һ��������ϣ���һ�������·�Ӧת��Ϊ��Ҫ�Ļ���ԭ����ϩ[C2H4]���仯ѧ����ʽ��д����2CO2+6H2�TC2H4+4H2O����6gH2ת��CO2�����ܴ�����״���µ�CO2���������������֪����״���£�CO2���ܶ�Ϊ1.977g/L������������1λС����
���𰸡���������1������̼���ָ����������Ϣʱ�����õ�����Ҫ�������٣��ر��Ǽ��ٶ�����̼���ŷ�����������̬�����Դӽڵ硢���ܺͻ��յȻ������ı�����ϸ�ڣ��ݴ˽��з�����ɣ�
��2�����ݲμӷ�Ӧ�������������ɷ�Ӧ�Ļ�ѧ����ʽ������ܴ�����״���µĶ�����̼����������������ö�����̼�������
����⣺��1����ʹ��һ����ľ��ɿ��Լ���ɭ�ֵĿ�������ľ����ͨ������������ո���Ķ�����̼���������ֹصƣ��ܽ�Լ���ܣ����ٶ�����̼���ŷţ������ϡ���̼������
��2�����ܴ�����״���µĶ�����̼������Ϊx��
2CO2+6H2=C2H4+4H2O
88 12
x 6g
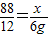 x=44g
x=44g
�ܴ�����״���µĶ�����̼�����=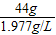 =22.3L��
=22.3L��
�𣺣�1����ʹ��һ����ľ��ȣ��������ɣ���
��2���ܴ�����״���µ�CO2����22.3L��
�����������ѶȲ������ո��ݻ�ѧ����ʽ�ļ��㼴����ȷ����⣬����ʱҪע�����Ĺ淶�ԣ�
��2�����ݲμӷ�Ӧ�������������ɷ�Ӧ�Ļ�ѧ����ʽ������ܴ�����״���µĶ�����̼����������������ö�����̼�������
����⣺��1����ʹ��һ����ľ��ɿ��Լ���ɭ�ֵĿ�������ľ����ͨ������������ո���Ķ�����̼���������ֹصƣ��ܽ�Լ���ܣ����ٶ�����̼���ŷţ������ϡ���̼������
��2�����ܴ�����״���µĶ�����̼������Ϊx��
2CO2+6H2=C2H4+4H2O
88 12
x 6g
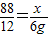 x=44g
x=44g�ܴ�����״���µĶ�����̼�����=
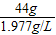 =22.3L��
=22.3L���𣺣�1����ʹ��һ����ľ��ȣ��������ɣ���
��2���ܴ�����״���µ�CO2����22.3L��
�����������ѶȲ������ո��ݻ�ѧ����ʽ�ļ��㼴����ȷ����⣬����ʱҪע�����Ĺ淶�ԣ�

��ϰ��ϵ�д�
 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д�
�����Ŀ
 2011��11��28����12��9�գ������Ϲ�����仯��ܹ�Լ����17�ε�Լ���������ϷǶ����������е°���У��й��������ڼ��ŵij�ŵ���ٴγ��չʾ���й�ı��չ���ٺ����������εĴ������Ŀǰ������̼������Ϊ����Ĺ�ʶ��
2011��11��28����12��9�գ������Ϲ�����仯��ܹ�Լ����17�ε�Լ���������ϷǶ����������е°���У��й��������ڼ��ŵij�ŵ���ٴγ��չʾ���й�ı��չ���ٺ����������εĴ������Ŀǰ������̼������Ϊ����Ĺ�ʶ�� 2011��11��28����12��11�գ����Ϲ�����仯��ܹ�Լ��17�ε�Լ���������Ϸǵ°��ٿ����������鶨�顷�ڶ���ŵ�ڵĴ������⣬�ǵ°����ڴ���������ؼ����⣮�ҹ���������ʼ��ֹ���ں�������ͨ�����׳����������ģ���ͼ������ǰ׳������ɣ����ͼ�ش��������⣮
2011��11��28����12��11�գ����Ϲ�����仯��ܹ�Լ��17�ε�Լ���������Ϸǵ°��ٿ����������鶨�顷�ڶ���ŵ�ڵĴ������⣬�ǵ°����ڴ���������ؼ����⣮�ҹ���������ʼ��ֹ���ں�������ͨ�����׳����������ģ���ͼ������ǰ׳������ɣ����ͼ�ش��������⣮