��Ŀ����
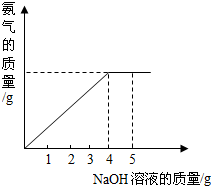
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O��Ӧ�����зų��İ�������������NaOH��Һ�������Ĺ�ϵ����ͼ��ʾ������㣺
��1��NH4NO3��N��H��O��������Ϊ______��
��2��ǡ����ȫ��Ӧ����NaOH��Һ������Ϊ______g��
��3����Ʒ������淋�����������
��4��ǡ����ȫ��Ӧʱ������Һ��NaNO3����������������һλС������

���𰸡���������1�������и�Ԫ�ص�������Ϊ��ԭ�ӵ����ԭ����������ԭ�ӵĸ����Ļ�֮�ȣ�
��2����ͼ�пɿ�������������Һ�μӵ�4��ʱ�����ǡ����ȫ��Ӧ��
��3�����ݲμӷ�Ӧ���������Ƶ������������淋����������� ×100%�����Ʒ������淋�����������
×100%�����Ʒ������淋�����������
��3�����ݲμӷ�Ӧ���������Ƶ�����������ɵ������Ƶ������������ɵ������Ƶ�����+ԭ�е������Ƶ�����Ϊ������Һ�������Ƶ�����������Ʒ������+��������������Һ������-�����������������������ɸ����������Ƶ����������Ϊ������Һ���������ٸ��� ×100%�������������Һ�������Ƶ�����������
×100%�������������Һ�������Ƶ�����������
����⣺��1��������е�Ԫ�ء���Ԫ�ء���Ԫ�ص�������=14×2��1×4��16×3=7��1��12
��2����ͼ��֪����������������������ʱ��˵������狀���������ǡ�÷�Ӧ������ǡ����ȫ��Ӧ����NaOH��Һ������Ϊ4g��
��3����2.0g��Ʒ��NH4NO3��������Ϊ������Ӧ����NaNO3������Ϊy����Ӧ���ɵ�NH3������Ϊz��
NH4NO3+NaOH�TNaNO3+NH3��+H2O
80 40 85 17
��4g×20% y z
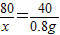
x=1.6g
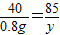
y=1.7g
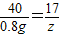
z=0.34g
��Ʒ������淋���������=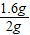 ×100%=80%
×100%=80%
��4��������Һ�������Ƶ�����=2g-1.6g+1.7g=2.1g
������Һ������=2g+5g-0.34g=6.66g
������Һ�������Ƶ���������=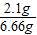 ×100%=31.5%
×100%=31.5%
�ʴ�Ϊ��
��1��7��1��12��
��2��4g��
��3��80%��
��4��31.5%��
��������������ʱҪ��4������������Һ��5������������Һ�����壬�������ã��μӷ�Ӧ������������Һ��������4�ˣ����ӵ�����������Һ��������5�ˣ�Ȼ�������û�ѧ��Ӧ����ʽ���м��㣮
��2����ͼ�пɿ�������������Һ�μӵ�4��ʱ�����ǡ����ȫ��Ӧ��
��3�����ݲμӷ�Ӧ���������Ƶ������������淋�����������
 ×100%�����Ʒ������淋�����������
×100%�����Ʒ������淋�������������3�����ݲμӷ�Ӧ���������Ƶ�����������ɵ������Ƶ������������ɵ������Ƶ�����+ԭ�е������Ƶ�����Ϊ������Һ�������Ƶ�����������Ʒ������+��������������Һ������-�����������������������ɸ����������Ƶ����������Ϊ������Һ���������ٸ���
 ×100%�������������Һ�������Ƶ�����������
×100%�������������Һ�������Ƶ���������������⣺��1��������е�Ԫ�ء���Ԫ�ء���Ԫ�ص�������=14×2��1×4��16×3=7��1��12
��2����ͼ��֪����������������������ʱ��˵������狀���������ǡ�÷�Ӧ������ǡ����ȫ��Ӧ����NaOH��Һ������Ϊ4g��
��3����2.0g��Ʒ��NH4NO3��������Ϊ������Ӧ����NaNO3������Ϊy����Ӧ���ɵ�NH3������Ϊz��
NH4NO3+NaOH�TNaNO3+NH3��+H2O
80 40 85 17
��4g×20% y z
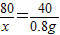
x=1.6g
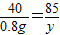
y=1.7g
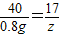
z=0.34g
��Ʒ������淋���������=
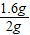 ×100%=80%
×100%=80% ��4��������Һ�������Ƶ�����=2g-1.6g+1.7g=2.1g
������Һ������=2g+5g-0.34g=6.66g
������Һ�������Ƶ���������=
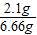 ×100%=31.5%
×100%=31.5%�ʴ�Ϊ��
��1��7��1��12��
��2��4g��
��3��80%��
��4��31.5%��
��������������ʱҪ��4������������Һ��5������������Һ�����壬�������ã��μӷ�Ӧ������������Һ��������4�ˣ����ӵ�����������Һ��������5�ˣ�Ȼ�������û�ѧ��Ӧ����ʽ���м��㣮

��ϰ��ϵ�д�
 �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д� ����ѧ��Ӧ�����ϵ�д�
����ѧ��Ӧ�����ϵ�д�
�����Ŀ
 �������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ����ͼ��ʾ��
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ����ͼ��ʾ��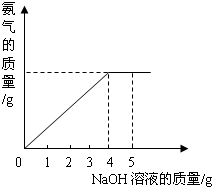 �������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��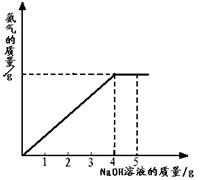 �������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��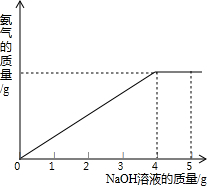 �������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����20%��NaOH��Һ���������·�Ӧ��
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij��������NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ 2.0g ���������Ʒ�������У�����20%��NaOH��Һ���������·�Ӧ�� �������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij�������� NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ2.0g���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��������йؼ��㣺
�������ũҵ�����г��õĻ�ѧ���ϣ�Ϊ�ⶨij�������� NaNO3���������Ʒ��NH4NO3�Ĵ��ȣ�ȡ2.0g���������Ʒ�������У�����5.0g 20%��NaOH��Һ���������·�Ӧ��NH4NO3+NaOH=NaNO3+NH3��+H2O����Ӧ�����зų��İ��������������NaOH��Һ�������Ĺ�ϵ��ͼ��ʾ��������йؼ��㣺