��Ŀ����
��1����ѧ��ȤС���ͬѧ�ڲⶨij������ʯ��������������������ͬѧ��ȡ��ʯ��Ʒ10g����������ϡ���ᣬ��ȫ��Ӧ��Fe2O3+6HCl=2FeCl3+3H2O��������ȥϡ����109.5g���˵õ�����2g ���������ʼȲ�����ˮҲ�����ᷢ����Ӧ��������������ʯ�������������������ͷ�Ӧ����Һ�����ʵ���������������д��������̣�����2��������ʯ������������������������������������______����ߡ��͡�����ԭ���ǣ�______��
���𰸡���������1���������֪������ʯ�е����ʼȲ������ᣬҲ������ˮ�����Ҹ���Ŀ���漰�Ļ�ѧ��ӦҲ�������ɳ����������������������dz�����ʯ�����ʵ���������ô��Ʒ��Fe2O3������=����Ʒ������-���ʵ�������������ʯ������������������= ×100%��
×100%��
�������⣬��Ӧ��������FeCl3�����Ը��ݻ�ѧ�������������������Ӧ��������Һ������=��Ӧǰ�������ʵ�����-���ʵ����������ݷ�Ӧ��������Һ�����ʵ���������= ×100%���������Ӧ����Һ�����ʵ�����������
×100%���������Ӧ����Һ�����ʵ�����������
��2��������ʯ�Ǻ������ʵ����������ݴ˼����ж��������������ĸߵͣ�
����⣺��1�����������֪����������������������Ϊ��10g-2g=8g
��������������������������ǣ�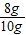 ×100%=80.0%��
×100%=80.0%��
�跴Ӧ������FeCl3������Ϊx
Fe2O3+6HCl�T2FeCl3+3H2O
160 325
8g x
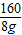 =
=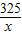
��ã�x=16.25g
��Ӧ������FeCl3��Һ������Ϊ��10g+109.5g-2g=117.5g��
��Ӧ������FeCl3��Һ����������Ϊ��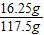 ×100%=13.8%��
×100%=13.8%��
��2������ʯ�г��˺��������������������������ʣ�
�ʳ�����ʯ�������������������������������������ͣ�
�𣺣�1����������������������������80%����Ӧ��������Һ�����ʵ���������Ϊ13.8%��
��2��������ʯ�������������������������������������ͣ�ԭ��������ʯ�г��˺��������������������������ʣ�
����������һ�������Ļ�ѧ�ۺϼ����⣮����Ĺؼ�����Ū�廯ѧ��Ӧ�з�Ӧ�������֮��Ĺ�ϵ��ͬʱҪŪ�������ڷ�Ӧ�д�����ʲô�ط����ڶ����й��ڷ�Ӧ����Һ�����ʵ����������ļ��㣬�ؼ���ҪŪ�巴Ӧ����Һ��ʲô��Һ��һ����˵���ɸ��ݻ�ѧ����ʽ�����ʵ���������Һ�������ɸ��ݹ�ʽ������Ӧ����Һ������=��Ӧǰ�����������������֮��-���ʵ�����-���������-�������������������
 ×100%��
×100%���������⣬��Ӧ��������FeCl3�����Ը��ݻ�ѧ�������������������Ӧ��������Һ������=��Ӧǰ�������ʵ�����-���ʵ����������ݷ�Ӧ��������Һ�����ʵ���������=
 ×100%���������Ӧ����Һ�����ʵ�����������
×100%���������Ӧ����Һ�����ʵ�������������2��������ʯ�Ǻ������ʵ����������ݴ˼����ж��������������ĸߵͣ�
����⣺��1�����������֪����������������������Ϊ��10g-2g=8g
��������������������������ǣ�
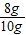 ×100%=80.0%��
×100%=80.0%���跴Ӧ������FeCl3������Ϊx
Fe2O3+6HCl�T2FeCl3+3H2O
160 325
8g x
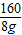 =
=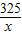
��ã�x=16.25g
��Ӧ������FeCl3��Һ������Ϊ��10g+109.5g-2g=117.5g��
��Ӧ������FeCl3��Һ����������Ϊ��
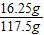 ×100%=13.8%��
×100%=13.8%����2������ʯ�г��˺��������������������������ʣ�
�ʳ�����ʯ�������������������������������������ͣ�
�𣺣�1����������������������������80%����Ӧ��������Һ�����ʵ���������Ϊ13.8%��
��2��������ʯ�������������������������������������ͣ�ԭ��������ʯ�г��˺��������������������������ʣ�
����������һ�������Ļ�ѧ�ۺϼ����⣮����Ĺؼ�����Ū�廯ѧ��Ӧ�з�Ӧ�������֮��Ĺ�ϵ��ͬʱҪŪ�������ڷ�Ӧ�д�����ʲô�ط����ڶ����й��ڷ�Ӧ����Һ�����ʵ����������ļ��㣬�ؼ���ҪŪ�巴Ӧ����Һ��ʲô��Һ��һ����˵���ɸ��ݻ�ѧ����ʽ�����ʵ���������Һ�������ɸ��ݹ�ʽ������Ӧ����Һ������=��Ӧǰ�����������������֮��-���ʵ�����-���������-�������������������

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
 ��2007?���£�ijУ��ѧ��ȤС���ѧУ������ˮ��״����չ��һϵ�е�ʵ�����о���������ͬѧ�ǵIJ���ʵ�����ݣ���������ǵ��о����ش��������⣺
��2007?���£�ijУ��ѧ��ȤС���ѧУ������ˮ��״����չ��һϵ�е�ʵ�����о���������ͬѧ�ǵIJ���ʵ�����ݣ���������ǵ��о����ش��������⣺


