��Ŀ����
�±��г��˲��������к����϶�Ԫ�ص�ԭ�ӽṹʾ��ͼ����ݱ��ش��������⣺
| H | O | Na | Mg | Cl | K | Ca |
 | 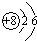 | 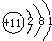 | 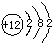 | 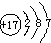 | 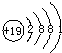 |  |
��2��ѡ������ʵ���Ԫ�أ����һ�ַ����������ݵ����ʣ����û�ѧʽ��ʾ�����������Ⱦ�ĵ���ȼ�ϣ�______��������ʵ������ȡ�������Σ�______��
��3���ɱ��в���Ԫ����ɵ���������֮���ͨ���кͷ�Ӧ�����Ȼ�þ���仯ѧ��Ӧ����ʽΪ______��
�⣺��1����ԭ�Ӻ������ĸ����Ӳ㣬���������ڵ��Ӳ������ʸ�Ԫ���ڵ������ڣ�������ȱ���ù������ɣ���ԭ������������������ӣ���ʧȥ�������ӣ�ʹ������Ϊ�����ﵽ8�����ȶ��ṹ������Ϊ Ca2+
��2���������Ⱦ��ȼ������������ѧʽΪH2������Ԫ����ɵ����������������ʵ��������������ѧʽΪKClO3
��3���кͷ�Ӧ������Ӧ�����κ�ˮ�ķ�Ӧ��Ҫ�����Ȼ�þ�����������ᣬ����������þ����ѧ��Ӧ����ʽΪ2HCl+Mg��OH��2=MgCl2+2 H 2O
�ʴ�Ϊ����1��4 ��������֢ Ca2+��2����H2��KClO3��3��2HCl+Mg��OH��2=MgCl2+2 H 2O
��������1���������������ڵ��Ӳ���������������С��4����ʧȥ���ӳ�Ϊ�����ӣ��������ӷ��ŵ���д������д���ӷ���
��2��������ĿҪ��ͻ�ѧʽ��д��д������Ҫ��Ļ�ѧʽ��
��3�������кͷ�Ӧ�Ķ���ͻ�ѧ����ʽ����дԭ����д��ѧ����ʽ��
��������Ҫ������ԭ�ӵĽṹʾ��ͼ����ص�֪ʶ������ѧ����������������
��2���������Ⱦ��ȼ������������ѧʽΪH2������Ԫ����ɵ����������������ʵ��������������ѧʽΪKClO3
��3���кͷ�Ӧ������Ӧ�����κ�ˮ�ķ�Ӧ��Ҫ�����Ȼ�þ�����������ᣬ����������þ����ѧ��Ӧ����ʽΪ2HCl+Mg��OH��2=MgCl2+2 H 2O
�ʴ�Ϊ����1��4 ��������֢ Ca2+��2����H2��KClO3��3��2HCl+Mg��OH��2=MgCl2+2 H 2O
��������1���������������ڵ��Ӳ���������������С��4����ʧȥ���ӳ�Ϊ�����ӣ��������ӷ��ŵ���д������д���ӷ���
��2��������ĿҪ��ͻ�ѧʽ��д��д������Ҫ��Ļ�ѧʽ��
��3�������кͷ�Ӧ�Ķ���ͻ�ѧ����ʽ����дԭ����д��ѧ����ʽ��
��������Ҫ������ԭ�ӵĽṹʾ��ͼ����ص�֪ʶ������ѧ����������������

��ϰ��ϵ�д�
 ��ѧʵ����ϵ�д�
��ѧʵ����ϵ�д�
�����Ŀ






















