��Ŀ����
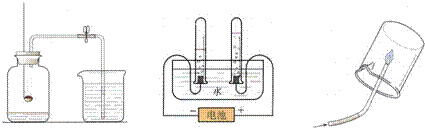
С���ü�������������ʵ�飺����������ϡ�����ϣ�������������ٽ����ɵ�����ͨ�뵽����ʯ��ˮ�У�����ʯ��ˮ����ǣ�����ش�
��1�����ƶϲ�����������
��2����������
��3��������������ʯ��ˮ��Ӧ�Ļ�ѧ����ʽΪ
��1�����ƶϲ�����������
CO2
CO2
�����������CO32-
CO32-
���ӣ���2����������
�����
�����
���������������3��������������ʯ��ˮ��Ӧ�Ļ�ѧ����ʽΪ
CO2+Ca��OH��2=CaCO3��+H2O
CO2+Ca��OH��2=CaCO3��+H2O
����������1�����ݷ�Ӧ�����ж�������ͷ�Ӧ��ijɷ֣�������̼��ʹ����ʯ��ˮ����ǣ�̼�����ܺ����ᷴӦ���ɶ�����̼���壻
��2�����ݼ����ǵ���ɳɷ��жϣ�
��3�����ݷ�Ӧ�����������д��ѧ����ʽ��ע����ƽ��������
��2�����ݼ����ǵ���ɳɷ��жϣ�
��3�����ݷ�Ӧ�����������д��ѧ����ʽ��ע����ƽ��������
����⣺��1���������к���̼��ƣ����Ե��μ�����ʱ�������̼��Ʒ�Ӧ�����ɶ�����̼��������̼��ʹ�����ʯ��ˮ����ǣ�
��2���������г�̼���������������ʣ����ڻ���
��3��������̼�����ʯ��ˮ��Ӧ����̼��Ƴ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ CO2+Ca��OH��2=CaCO3��+H2O��
�ʴ�Ϊ����1��CO2��CO32-��
��2������
��3��CO2+Ca��OH��2=CaCO3��+H2O
��2���������г�̼���������������ʣ����ڻ���
��3��������̼�����ʯ��ˮ��Ӧ����̼��Ƴ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ CO2+Ca��OH��2=CaCO3��+H2O��
�ʴ�Ϊ����1��CO2��CO32-��
��2������
��3��CO2+Ca��OH��2=CaCO3��+H2O
������̼������ӵļ��飬����ֻ�����ᣮ��ֻ�����������������һ�����ж�����̼���壬���Ա�����֤���õ������Ƕ�����̼������˵������̼�����

��ϰ��ϵ�д�
 ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д�
ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д�
�����Ŀ