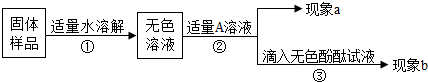
��Ŀ����
ij��ѧ�С���ͬѧ��Ϊ�˲ⶨ��ʡһ��ֽ���ŷŵ���ˮ���������Ƶĺ����������Ƚ�������������Ϊ3 7%���ܶ�1.18g/cm3��Ũ����ϡ�ͳ�������������Ϊ5%��ϡ����200g��Ȼ��ȡ80��Og��ˮ���ձ��У���μ������ƺõ�ϡ����7 3��Og��ǡ����ȫ��Ӧ����������ˮ�е������ɷֲ������ᷴӦ��������㣺��1������ϡ����ʱ����ȡ��������������Ϊ3 7%��Ũ������ٺ�����
��2����ˮ�������������Ƶ����������Ƕ��٣�
���𰸡���������1��������Һϡ��ǰ�����ʵ�������������ɸ���Ľ��
��2������������������Ʒ�Ӧ�Ļ�ѧ����ʽ���������ĵ��������������������������Ƶ��������������Լ��������������Һ������������
����⣺��1���ݣ��������ǰ����ȣ�����ȡ��������������Ϊ3 7%��Ũ��������ΪV����
V?1.18g/cm3×37%=200g×5%
��ã�V=22.9mL
��2�����ĵ�73.0g�����к��Ȼ��������Ϊ��73.0g×5%=3.65g��
����ˮ�к���NaOH����Ϊx��
HCl+NaOH�TNaCl+H2O
36.5 40
3.65g x
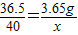
��ã�x=4g
������ˮ�������������Ƶ����������ǣ�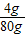 ×100% w=5%
×100% w=5%
�𣺣�1������ϡ����ʱ����ȡ��������������Ϊ3 7%��Ũ����22.9������
��2����ˮ�������������Ƶ�����������5%��
������������Ҫ�����˸��ݻ�ѧ����ʽ���е��йؼ��㣬���ʱ����������ʵĹ�ϵ��ȷ����д��ѧ����ʽ����������ϵ���б���ʽ���㼴�ɣ�
��2������������������Ʒ�Ӧ�Ļ�ѧ����ʽ���������ĵ��������������������������Ƶ��������������Լ��������������Һ������������
����⣺��1���ݣ��������ǰ����ȣ�����ȡ��������������Ϊ3 7%��Ũ��������ΪV����
V?1.18g/cm3×37%=200g×5%
��ã�V=22.9mL
��2�����ĵ�73.0g�����к��Ȼ��������Ϊ��73.0g×5%=3.65g��
����ˮ�к���NaOH����Ϊx��
HCl+NaOH�TNaCl+H2O
36.5 40
3.65g x
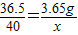
��ã�x=4g
������ˮ�������������Ƶ����������ǣ�
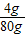 ×100% w=5%
×100% w=5%�𣺣�1������ϡ����ʱ����ȡ��������������Ϊ3 7%��Ũ����22.9������
��2����ˮ�������������Ƶ�����������5%��
������������Ҫ�����˸��ݻ�ѧ����ʽ���е��йؼ��㣬���ʱ����������ʵĹ�ϵ��ȷ����д��ѧ����ʽ����������ϵ���б���ʽ���㼴�ɣ�

��ϰ��ϵ�д�
�����Ŀ