��Ŀ����
������ԭ����ͭʵ��õ��Ĺ����ĩΪ����ͭ��ͭ�Ļ�����ѧ��ȤС���ͬѧ�Ը÷�ĩ�еĵ���ͭ�ĺ������вⶨ������ͬѧ��ȡ10g��ĩ��Ʒ�����ձ��У�Ȼ����벻ͬ������ͬŨ�ȵ�ϡ���ᣬ��ַ�Ӧ���ձ���ʣ�������г�����ʵ�����ݼ�¼���£�| ��� | ��Ʒ������g�� | ����ϡ����������g�� | ʣ�����������g�� |
| 1 | 10 | 20 | 8.4 |
| 2 | 10 | 30 | 8 |
| 3 | 10 | 40 | 8 |
���𰸡�����������ͼʾ���ݿ�֪���������������������ټ��٣�˵��ʣ������ʶ���ͭ���ɴ˿�֪����ͭ������������������ͭ�����������������������������������������������ʱҪ��������ȫ���μӷ�Ӧ�ĵ�һ�ν��м��㣮
����⣺���ݼ���30g����ͼ���40g����ʣ������������䣬˵��ʣ��������Ѿ�����������ͭ��ȫ��Ϊͭ������ͭ������Ϊ8g��
���ݵ�һ��ͬѧ�͵ڶ���ͬѧ���ݿ�֪����һ��ͬѧʵ��������ȫ���μ��˷�Ӧ������������ͭ����Ϊ��10g-8.4g=1.6g��������λͬѧ����������ͬ���ʿ��Ը��ݵ�һλͬѧ�IJ������ݼ��㣻
��20gϡ�����е�����H2SO4������Ϊx
CuO+H2SO4�TCuSO4+H2O
80 98
1.6g x
 =
=
x=1.96g
����ϡ�������ʵ����������ǣ� ×100%=9.8%
×100%=9.8%
�𣺼����ϡ����������������Ϊ9.8%��
�����������ؼ���Ҫ��������һ��ͬѧ��ʵ��ʱ����ȫ���μ��˷�Ӧ������ͭ��ʣ�࣬��������ͭ��������������������ټ�����������������
����⣺���ݼ���30g����ͼ���40g����ʣ������������䣬˵��ʣ��������Ѿ�����������ͭ��ȫ��Ϊͭ������ͭ������Ϊ8g��
���ݵ�һ��ͬѧ�͵ڶ���ͬѧ���ݿ�֪����һ��ͬѧʵ��������ȫ���μ��˷�Ӧ������������ͭ����Ϊ��10g-8.4g=1.6g��������λͬѧ����������ͬ���ʿ��Ը��ݵ�һλͬѧ�IJ������ݼ��㣻
��20gϡ�����е�����H2SO4������Ϊx
CuO+H2SO4�TCuSO4+H2O
80 98
1.6g x
 =
=
x=1.96g
����ϡ�������ʵ����������ǣ�
 ×100%=9.8%
×100%=9.8% �𣺼����ϡ����������������Ϊ9.8%��
�����������ؼ���Ҫ��������һ��ͬѧ��ʵ��ʱ����ȫ���μ��˷�Ӧ������ͭ��ʣ�࣬��������ͭ��������������������ټ�����������������

��ϰ��ϵ�д�
�����Ŀ

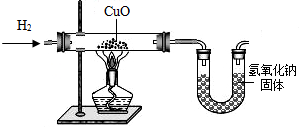
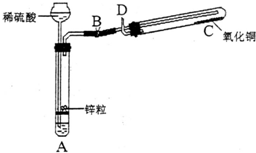 ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�
ijѧ������ͼ��ʾװ���Ʊ������������Ƶõ�������ԭ����ͭ��װ���б�Ҫ������̨�����С������豸����ͼ�о�����ȥ��������д���пհף�