��Ŀ����
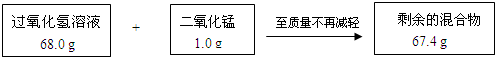
ij��ȤС��Ϊ�ⶨʵ������һƿ���õĹ���������Һ�����ʵ�����������ʵ���������������ͼ��ʾ��

��1��ʣ��Ļ�����ж������̵�����Ϊ g����Ӧ��������������Ϊ g��
��2������ù���������Һ����������������
��3��ʵ���������ͭ��Һ�ڸ÷�Ӧ��Ҳ���������̵����á�����5.6g��ˮ����
ͭ����������̽��б�ʵ�飬����ʣ�����Һ�����ʵ�����������������ͭȫ������ˮ������ʱ������0.1%��
��1��1.0�� 1.6�� ��������������������������ÿ��1�֣���2�֣�
��2����68.0 g����������Һ�к��й������������Ϊx��
2H2O22 H2O + O2�� ��1�֣�
68 32
x 1.6 68��32 = x ��1.6 ��1�֣�
x = 3.4g ��1�֣�
����������Һ�����ʵ���������= 3.4g/68.0g ��100% = 5.0% (1��)
��3������ͭ��Һ��������������= 5.6g / (67.4g�� 1.0g + 5.6g) ��100% ��1�֣�
�� 7.8% ��1�֣�
����:��