��Ŀ����
��5�֣���������Ҫ�Ľ������ϡ�
��1����������Ʒ����;�У����ý��������Ե��� ������ĸ����ͬ����


A����ǯ B������ C��ˮ��ͷ
��2��Ŀǰ����������50%���ϵķϸ����õ��������ã���Ŀ���� ��
A����Լ������Դ B���������ɿ��� C����ֹ��������
��3��ˮ��ͷ�������һ�������Ϊ��ֹ�����⡣��д��һ�ַ�ֹѧУ��������ķ��� ��
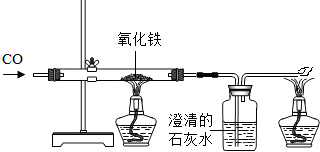
��4����ҵ����һ����̼�ͳ�������Ҫ�ɷ�����������ұ��������Ӧ�Ļ�ѧ����ʽΪ____��
��5�������צ���Fe�ۡ���Ҳ��������ʳƷ���ʣ���֮Ϊ��˫�����������ܹ����տ����е�ˮ�ֺ����������û�ѧ��������ʹ��һ��ʱ���ġ�˫�������Ƿ���ȫʧЧ��д��������� ��
��1��B ��2��A ��3��Ϳ��
��4��3CO+Fe2O3 ���� 2Fe+3CO2
��5��ȡ������˫���������Թ��У��ڼ���ϡ���ᣬ�۲쵽��Һ����ɫ��ɻ�ɫ���������ݷų�����֤��˫������Ч����ֻ����Һ����ɫ��ɻ�ɫ�������ݷų�����˫������Ч��
���������������1��������Ӳ��һ��Ƚϴ������õĵ����ԡ������ԡ���չ�Եȣ������������������������������õĵ����ԣ�
��2���Ͼɽ����Ļ������ã��ȿ��Խ�Լ��Դ���ֿ��Ա���������
��3�������������������֪��ֻҪʹ����������ˮ�����Ϳ��Է�ֹ���⣻��Ϳ����Է�ֹѧУ�������⣻
��4�����ݷ�Ӧ��������P�������غ㶨�ɿ�����д��ѧ����ʽ����3CO+Fe2O3����2Fe+3CO2��
��5�����������֪��֤����˫�������Ƿ���ȫʧЧ������֤����˫���������Ƿ����������û�ѧ��������ʹ��һ��ʱ���ġ�˫�������Ƿ���ȫʧЧ�ķ����ǣ�ȡ������˫���������Թ��У��ټ���ϡ���ᣬ���۲쵽��Һ����ɫ��ɻ�ɫ���������ݷų�����֤��˫������Ч����ֻ����Һ����ɫ��ɻ�ɫ�������ݷų�����˫������Ч��
���㣺�������������ʼ���;��ʳƷ����������ʼ�����հ�װ�ijɷ�̽����һ����̼��ԭ��������������ʴ��������������������Ļ������ü�����Ҫ�ԣ���д��ѧ����ʽ
�����������ѶȲ��Ǻܴ��漰֪ʶ��϶ࡢ�ۺ��Խ�ǿ�����ս��������ʡ���ѧ����ʽ����д�����ܻ����ȷ������������ȷ�����Ĺؼ�������ע�⣬���ʱҪ���������ܻ�������Ҫ�ԣ�Ҫ���������غ㶨������ȷ����д��ѧ����ʽ��

 �Ͻ�ƽ��У����ϵ�д�
�Ͻ�ƽ��У����ϵ�д�
 ��������Ҫ�Ľ������ϣ��ڽ��컴����ˮ�ɻ���ʱ�������˴����ĸ�����
��������Ҫ�Ľ������ϣ��ڽ��컴����ˮ�ɻ���ʱ�������˴����ĸ�����