��Ŀ����
ijͬѧ���ⶨ���������ƺ�ζ����ʳ�εĺ���������������ʵ�飺
�ٳ�ȡ���ƺŵ�ζ����Ʒ5.00�ˣ�����������ˮ��
�ڼ�����������������Һ���ٵμ�ϡ���ᣮ
�۹��ˣ�������ϴ�ӡ���ɡ��������õ���������Ϊ1.44�ˣ�
��������ʵ�鲽�輰ͼʾװ�ûش��������⣺
��1�����˲�������IJ���������______��
��2�����������Ļ�ѧ��Ӧ����ʽΪ______��
��3����ζ����װ�ϱ�ע����NaCl������10%���������Ʒ______
������ϡ������ϡ�����Ʒ��ʾ������������
�ٳ�ȡ���ƺŵ�ζ����Ʒ5.00�ˣ�����������ˮ��
�ڼ�����������������Һ���ٵμ�ϡ���ᣮ
�۹��ˣ�������ϴ�ӡ���ɡ��������õ���������Ϊ1.44�ˣ�
��������ʵ�鲽�輰ͼʾװ�ûش��������⣺
��1�����˲�������IJ���������______��
��2�����������Ļ�ѧ��Ӧ����ʽΪ______��
��3����ζ����װ�ϱ�ע����NaCl������10%���������Ʒ______
������ϡ������ϡ�����Ʒ��ʾ������������

��1���ڽ��й��˲���ʱ������ʹ������̨�⣬��Ҫ�õ��ձ���©�����������Ȳ������������в��������������ã���2��ʳ���к��������ӣ�������������Һ�������Ӿ��������ӽ�����ɰ�ɫ�Ȼ��������������Ļ�ѧ��ӦΪ��
NaCl+AgNO3=AgCl��+NaNO3��
��3��������������Ϊ�˽��Ȼ����е���ת��Ϊ�ɳ������Ȼ���������ϡ�����Ŀ���ǽ��������ʲ����ij����ܽ��ȥ��
����Ʒ���Ȼ��Ƶ�����Ϊx������ݷ���ʽ�У�
NaCl+AgNO3=AgCl��+NaNO3
58.5 144
x 1.44g
x=
=0.585g
������Ʒ���Ȼ��Ƶĺ���Ϊ��
��100%=11.7%
��11.8%��10%���ʴ���Ʒ�����ϲ�Ʒ��ʾ������������
�ʴ�Ϊ��
��1���ձ���©������������
��2��NaCl+AgNO3=AgCl��+NaNO3��
��3�������ϣ�
NaCl+AgNO3=AgCl��+NaNO3��
��3��������������Ϊ�˽��Ȼ����е���ת��Ϊ�ɳ������Ȼ���������ϡ�����Ŀ���ǽ��������ʲ����ij����ܽ��ȥ��
����Ʒ���Ȼ��Ƶ�����Ϊx������ݷ���ʽ�У�
NaCl+AgNO3=AgCl��+NaNO3
58.5 144
x 1.44g
x=
| 58.5��1.44 |
| 144 |
������Ʒ���Ȼ��Ƶĺ���Ϊ��
| 0.585 |
| 5 |
��11.8%��10%���ʴ���Ʒ�����ϲ�Ʒ��ʾ������������
�ʴ�Ϊ��
��1���ձ���©������������
��2��NaCl+AgNO3=AgCl��+NaNO3��
��3�������ϣ�

��ϰ��ϵ�д�
 ��һ������ĩ�ٷֳ�̾�ϵ�д�
��һ������ĩ�ٷֳ�̾�ϵ�д�
�����Ŀ
 ijͬѧ���ⶨ���������ƺ�ζ����ʳ�εĺ���������������ʵ�飺
ijͬѧ���ⶨ���������ƺ�ζ����ʳ�εĺ���������������ʵ�飺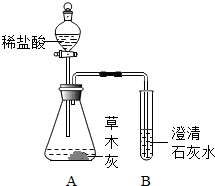 ��2012?�γ�ģ�⣩С��ͬѧ����������ʱ��С�Ľ�һƿ�״������ڻ�¯�Ե�һ�Ѳ�ľ���ϣ������д������������ɣ���������Ȥ��������������ѧϰС���ͬѧ���Բ�ľ�ҵijɷֽ���̽����
��2012?�γ�ģ�⣩С��ͬѧ����������ʱ��С�Ľ�һƿ�״������ڻ�¯�Ե�һ�Ѳ�ľ���ϣ������д������������ɣ���������Ȥ��������������ѧϰС���ͬѧ���Բ�ľ�ҵijɷֽ���̽����