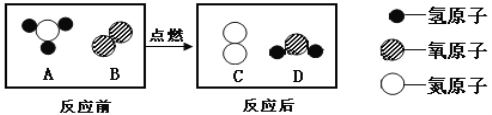
��Ŀ����
��ȤС���ͬѧ���������֪ʶ����̽����
��֪ʶ�عˣ�
��1�����ǵؿ��к������Ľ���Ԫ�أ�����Ȼ����Ҫ�Ի���̬����ʽ�������������С�����˵������ȷ���� _____��

A ��ԭ�ӵ�������Ϊ13
B �����Ӻ������������Ӳ�
C ����������Ԫ��ͨ����+3��
D �������������������������õĵ�����
������ұ����
��ҵ�ϳ�����������Ҫ�ɷ�ΪAl2O3����Fe2O3���ʣ�Ϊԭ��ұ����������������ͼ����������������������ᡢ��ܷ�����Ӧ��Al2O3��2NaOH��2NaAlO2��H2O����ش��������⣺

��2���Լ�X��_________ѡ������������Һ�����ᣩ��
��3����Ӧ�ٺ�������õij�����___________��
��4��д����Ӧ�ڵĻ�ѧ����ʽ___________________��
���������

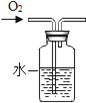
��������⣩��ͼ��ʾװ��������ϡ���ᷴӦ������ȡ��������ô���ܷ���ˮ��Ӧ���������أ�
���������ϣ�
���� | ����ˮ | ����ˮ | ����ˮ |
�ܷ�����Ӧ | �ܷ�Ӧ | �ܷ�Ӧ | ����Ӧ |
�����������ۣ�
��5�����ڿ�������������Ӧ�����������һ�����ܵ���������Ĥ������Ĥ�����á��������п���������ˮ������������Ӧ�Ļ�ѧ����ʽΪ_____________��
��6����ȥ��������������Ĥ�ķ����У���һ����_______�������������⣬�����Խ���Ƭ�����Ȼ���( HgCl2)��Һ�У��γ������루�����Ͻ��ƻ����������ܵ�����Ĥ�ҷ�ֹ���������ɡ�
��ʵ��̽����
��7������Ƭ��ȫ�����Ȼ���(HgCl2)��Һ��1�������ң�ȡ������������ˮ��ϴ�ɾ�����뵽����ˮ�У���ͼ������������ɴ������ݡ���Χ���ְ�ɫ���������رյ����ϵĻ���K����Ӧֹͣ����ԭ����_____________��
��8���ڵ��ܿ�a��Ϳ����ˮ�����ֵ��ܿ��������γɣ��������뿪���ܺ�����______����³�����������������ȼ�ŵ�ľ����ȼ���ݣ�������������
��ʵ����ۣ�
��9��������ˮ��Ӧ����������д������ˮ��Ӧ�������������������Ļ�ѧ����ʽ____________��
�������о�����ȤС��ȡ8g��þ�Ͻ���Ʒ����30g����������Һƽ���ֳ��������μ�����Ʒ�У�þ������������Һ����Ӧ����������������Һ��Ӧ�Ļ�ѧ����ʽΪ��2Al+2NaOH+2H2O�T2NaAlO2+3H2��������ַ�Ӧ���˳����壬����ϴ�ӡ������������ʵ���������£�
��������������Һ�Ĵ��� | ��һ�� | �ڶ��� | ������ |
ʣ����������/g | 5.3 | 2.6 | 2.0 |
��10���úϽ��У�þ������������Ϊ _____��
��11����������������Һ�����ʵ���������Ϊ���٣���д��������̣�____
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�ijѧУ��ѧ��ȤС���ͬѧ���ڻ�ѧʵ���ҷ���һƿʯ��ˮ�Լ�ƿƿ�ں�ƿ����Χ��һЩ��ɫ��ĩ��Ϊ���������ɫ��ĩ�ijɷּ��γ�ԭ�����ǽ���������̽����
��������⣩��ɫ��ĩ�ijɷֺ��γ�ԭ����ʲô��
���������룩����1��������CaCO3 ����2��������Ca(OH)2 ����3����������______
��ʵ��̽���� ��ͬѧȡ��ɫ��ĩ���Թ��У���������ˮ���������Թܵ��а�ɫ���壬�������ó���ɫ��ĩ��CaCO3�Ľ��ۡ� ��ͬѧ��Ϊ��ͬѧ�Ľ��۲��Ͻ���������______
Ϊ�˵ó������ܵĽ��ۣ����ǽ����±���ʾʵ�飬�����ʵ�鱨��
ʵ�鲽�� | ʵ������ | ʵ����� |
��ȡ�����Թ��е��ϲ���Һ���μ�1-2����ɫ��̪��Һ | ��Һ�ʺ�ɫ | ��ɫ��ĩ�к���______ |
���������Թܲ����İ�ɫ�����м���______ | ______ | ��ɫ��ĩ�к���CaCO3 |
������ʵ���֪����ɫ��ĩ�ɷֵIJ���________����ȷ�ġ�
����չ��˼��ͨ������ʵ���̽����ͬѧ����ʵ��Ա����˱��桢ʹ���ױ�����Һ�����л���֪ʶ���飬�������� ______________________��
A �ܷⱣ�� B ʹ��ǰ��ʱ���� C ��ֹʹ�ø��Լ�
������װ�ý���ʵ�飬���ܴﵽʵ��Ŀ����
|
|
|
|
A����֤CO2���� | B��֤��HCl������ˮ | C������������� | D���ռ����� |
A. A B. B C. C D. D