��Ŀ����
�����еĸ�Ԫ����Ҫ�����ڹ����������У����ǻ������[Ca10(PO4)6(OH)2]��ʽ���ڣ�����Է�������Ϊ1 004��ţ�̺��Ʒḻ�������գ���ţ���иƺ��ױ������ʣ��ǽ��ǵ�����ʳƷ��
��ͼ��ij��ҵ��˾��ţ�̰�װ��ǩ�ϵIJ������֡�
����ϸ�Ķ���ش��������⣺
��1����װ��ǩ��֬����3.3 g,��ָ100 mLţ���У���֬������������Ϊ3.3 g . ��ôһ��ţ���к������� g(������0.01 g) ��
��2�����ǻ�������и�Ԫ�ص����������� ������1λС����
��3��������ÿ��������Ҫ0.6 g�ƣ�����Щ����90%����ţ�̣���һ����ÿ������Ҫ�ȶ��ٺ�ţ�̣�
��1��0.28 ��2��39.8% ��3��2
�������������
�⣺1���裺һ��ţ���к��Ƶ�����ΪX��
100��0.11=250��X
X=0.28g
2���ǻ�������и�Ԫ�ص���������=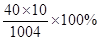 =39.8%��
=39.8%��
3���裺һ����ÿ������Ҫ��y��ţ�̡���
y��0.28��90%=0.6
y=2
�𣺣��ԣ�
���㣺Ԫ�ص�����������
������ijԪ�ص���������=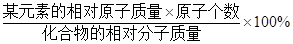 ��
��

��ϰ��ϵ�д�
 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д� �ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�ϴ�̸�������������νӽ̳��Ͼ���ѧ������ϵ�д�
�����Ŀ
�����еĸ�Ԫ����Ҫ�����ڹ����������У����ǻ�����ƾ���[Ca10��PO4��6��OH��2]��ʽ���ڣ�����Է�������Ϊ1004��ţ�̺��Ʒḻ�������գ���ţ�̵ĸƺ��ױ������ʣ��ǽ��ǵ�����ʳƷ����ͼ��ij��ҵ��˾��ţ�̰�װ��ǩ�IJ������֣�����ϸ�Ķ���ش��������⣺
��1����װ��ǩ��֬����3.3g����ָ100mLţ���У���֬������������Ϊ3.3g����ôһ��ţ�̺������� g��������0.01g����
��1����װ��ǩ��֬����3.3g����ָ100mLţ���У���֬������������Ϊ3.3g����ôһ��ţ�̺�������
| ��ţ�� ���ϣ���ţ�� �����ʣ�8���� ��������250mL/�� Ӫ���ɷ֣���ÿ100mL�� �ơ�0.11g ֬����3.3g �����ʡ�2.9g��2�����ǻ�������и�Ԫ�ص���������������Ϊ0.1%���� ��3��������ÿ��������Ҫ0.6g�ƣ�����Щ��90%����ţ�̣���һ����ÿ������Ҫ�ȶ��ٺ�ţ�̣� |
 �����еĸ�Ԫ����Ҫ�����ڹǸ�������У������ǻ�����ƾ���[Ca10��PO4��6��OH��2]��ʽ���ڣ�ţ�̺��Ʒḻ�������գ��ǽ��ǵ�����ʳƷ����ͼ��ij��ţ�̰�װ��ǩ�IJ������֣�����ϸ�Ķ��ش�
�����еĸ�Ԫ����Ҫ�����ڹǸ�������У������ǻ�����ƾ���[Ca10��PO4��6��OH��2]��ʽ���ڣ�ţ�̺��Ʒḻ�������գ��ǽ��ǵ�����ʳƷ����ͼ��ij��ţ�̰�װ��ǩ�IJ������֣�����ϸ�Ķ��ش� ��2009?�����������еĸ�Ԫ����Ҫ���ǻ�����ƾ���[Ca10��PO4��6��OH��2]��ʽ�����ڹ����������У���ͼ��ij��ҵ��˾��ţ�̰�װ��ǩ�IJ�������˵��������˵������ȷ���ǣ�������
��2009?�����������еĸ�Ԫ����Ҫ���ǻ�����ƾ���[Ca10��PO4��6��OH��2]��ʽ�����ڹ����������У���ͼ��ij��ҵ��˾��ţ�̰�װ��ǩ�IJ�������˵��������˵������ȷ���ǣ�������