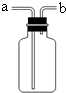
��Ŀ����
29�����������ʵ��װ��ͼ�ش��й����⣺

��1��ָ��ͼ�д�������������Ƣ�
��2��ѡ��Aװ����ȡ���壬�Է�Ӧ���״̬�ͷ�Ӧ������Ҫ����
��3��ijͬѧ��Bװ���ռ���һƿ������ˮ�����壬�������岻������ԭ�������
��4��Eװ�ÿɴ���Cװ���ռ����壬������Ӧ��

��1��ָ��ͼ�д�������������Ƣ�
����©��
��������ƿ
����2��ѡ��Aװ����ȡ���壬�Է�Ӧ���״̬�ͷ�Ӧ������Ҫ����
������Һ�巴Ӧ���������
����3��ijͬѧ��Bװ���ռ���һƿ������ˮ�����壬�������岻������ԭ�������
װ�������Բ��á��ռ�����ǰ����ƿ��δװ��ˮ��
����4��Eװ�ÿɴ���Cװ���ռ����壬������Ӧ��
b
��ͨ�루�a����b��������������1��ʶ��������
��2�����ݷ���װ�õ�ѡ������ɣ�
��3�����ɷ�������Ŀ����Ҫ���������
��4�������ռ�װ�õ��ռ�ԭ�����ɵõ���
��2�����ݷ���װ�õ�ѡ������ɣ�
��3�����ɷ�������Ŀ����Ҫ���������
��4�������ռ�װ�õ��ռ�ԭ�����ɵõ���
����⣺
��1��ʶ���������ʴ�Ϊ���ٳ���©���ڼ���ƿ
��2���۲�ͼ�β���Ϸ���װ�õ�ѡ�����֪���ʴ�Ϊ��������Һ�巴Ӧ���������
��3�����岻����˵��������п���������ֻҪ�п��ܵ��¿�����������ɶ����ԣ��ʴ�Ϊ��װ�������Բ��á��ռ�����ǰ����ƿ��δװ��ˮ��
��4��Cװ��˵�������ܶȱȿ���С�����������b�����ȳ�������ƿ�ϲ���������������࣬��ʹ�²������a��ȥ���ﵽ�ռ������Ŀ�ģ�
��1��ʶ���������ʴ�Ϊ���ٳ���©���ڼ���ƿ
��2���۲�ͼ�β���Ϸ���װ�õ�ѡ�����֪���ʴ�Ϊ��������Һ�巴Ӧ���������
��3�����岻����˵��������п���������ֻҪ�п��ܵ��¿�����������ɶ����ԣ��ʴ�Ϊ��װ�������Բ��á��ռ�����ǰ����ƿ��δװ��ˮ��
��4��Cװ��˵�������ܶȱȿ���С�����������b�����ȳ�������ƿ�ϲ���������������࣬��ʹ�²������a��ȥ���ﵽ�ռ������Ŀ�ģ�
������Ҫѧ�����շ���װ�õ��ռ�װ�õ�ѡ�����ͬʱ�����ռ������ԭ����

��ϰ��ϵ�д�
�����Ŀ