��Ŀ����
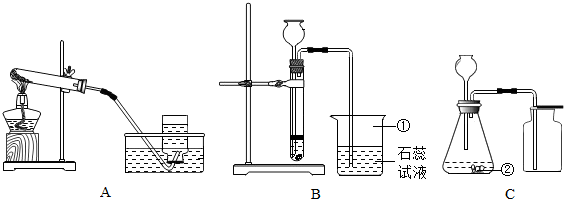
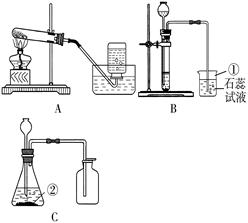
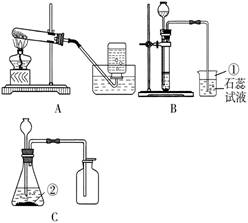
22��ij��ȤС��Ҫ�о�CO2������ЧӦ����ѡ�����к��ʵ�װ����գ�

��1��ʵ������ȡCO2�Ļ�ѧ����ʽΪ
��2��ʵ������ȡCO2��Ӧѡ
��3����ʵ������װ�â�ȥ������̼�������������Ȼ��⣬��������Ӧ��
��4��Ϊ�Ƚ϶�����̼�Ϳ���������ЧӦ���ɽ�ʢ������������ܱչ��ƿͬʱ���������½���ʵ�飮��ʵ���趨ʱ������������

��1��ʵ������ȡCO2�Ļ�ѧ����ʽΪ
CaCO3+2HCl�TCaCl2+H2O+CO2��
����2��ʵ������ȡCO2��Ӧѡ
��
������ţ���ͬ��������װ�ã�Ӧѡ��
���ռ�װ�ã���3����ʵ������װ�â�ȥ������̼�������������Ȼ��⣬��������Ӧ��
a
���a����b����������ͨ�룮��Ҫ�õ����﴿���Ķ�����̼���壬����װ��Ӧ����װ�â�֮��
���ǰ��������4��Ϊ�Ƚ϶�����̼�Ϳ���������ЧӦ���ɽ�ʢ������������ܱչ��ƿͬʱ���������½���ʵ�飮��ʵ���趨ʱ������������
�¶�
����������Ŀ������������װ�ú������ռ�װ�ã�ͬʱΧ�ƶ�����̼����ȡ�ռ������չ����Ŀ��ƣ�
��1����ȡ������̼���õ�ԭ����̼��ƺ�ϡ���ᣬ����д����ʽʱҪע�⣮
��2����ȡ������̼�����ǹ����Һ���ڲ����������½��У�����ѡ�â�װ������ȡ�����ڶ�����̼�ܽ���ˮ���ܶȱȿ���������ѡ�������ſ������ռ���ѡ�ܣ�
��3������������лӷ��ԣ����ŷ�Ӧ�Ľ��У��ռ��Ķ�����̼���������Ȼ������壬���Կ��Դ�ϴ��ƿ��֮��ȥ��ϴ��ƿ�dz����̳�������ȡ��������壬��Ҫ������װ�÷���ϴ��װ��֮���������Ա����������ˮ������
��4������ЧӦ�����Խ���ǵ�������嵼�µ��¶ȱ仯��������ʵ����Ӧ��ǿ������ע���¶ȵı仯�����
��1����ȡ������̼���õ�ԭ����̼��ƺ�ϡ���ᣬ����д����ʽʱҪע�⣮
��2����ȡ������̼�����ǹ����Һ���ڲ����������½��У�����ѡ�â�װ������ȡ�����ڶ�����̼�ܽ���ˮ���ܶȱȿ���������ѡ�������ſ������ռ���ѡ�ܣ�
��3������������лӷ��ԣ����ŷ�Ӧ�Ľ��У��ռ��Ķ�����̼���������Ȼ������壬���Կ��Դ�ϴ��ƿ��֮��ȥ��ϴ��ƿ�dz����̳�������ȡ��������壬��Ҫ������װ�÷���ϴ��װ��֮���������Ա����������ˮ������
��4������ЧӦ�����Խ���ǵ�������嵼�µ��¶ȱ仯��������ʵ����Ӧ��ǿ������ע���¶ȵı仯�����
����⣺��1����ȡ������̼���õ�ԭ����̼��ƺ�ϡ���ᣬ����д����ʽʱҪע�⣬����ʽΪCaCO3+2HCl�TCaCl2+H2O+CO2����
��2����ȡ������̼�����ǹ����Һ���ڲ����������½��У�����ѡ�â�װ������ȡ�����ڶ�����̼�ܽ���ˮ���ܶȱȿ���������ѡ�������ſ������ռ���ѡ�ܣ�
��3������������лӷ��ԣ����ŷ�Ӧ�Ľ��У��ռ��Ķ�����̼���������Ȼ������壬���Կ��Դ�ϴ��ƿ��֮��ȥ��ϴ��ƿ�dz����̳�������ȡ��������壬��Ҫ������װ�÷���ϴ��װ��֮���������Ա����������ˮ������
��4������ЧӦ�����Խ���ǵ�������嵼�µ��¶ȱ仯��������ʵ����Ӧ��ǿ������ע���¶ȵı仯�����
�ʴ�Ϊ��
��1��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���٣��ܣ�
��3��a����
��4���¶�
��2����ȡ������̼�����ǹ����Һ���ڲ����������½��У�����ѡ�â�װ������ȡ�����ڶ�����̼�ܽ���ˮ���ܶȱȿ���������ѡ�������ſ������ռ���ѡ�ܣ�
��3������������лӷ��ԣ����ŷ�Ӧ�Ľ��У��ռ��Ķ�����̼���������Ȼ������壬���Կ��Դ�ϴ��ƿ��֮��ȥ��ϴ��ƿ�dz����̳�������ȡ��������壬��Ҫ������װ�÷���ϴ��װ��֮���������Ա����������ˮ������
��4������ЧӦ�����Խ���ǵ�������嵼�µ��¶ȱ仯��������ʵ����Ӧ��ǿ������ע���¶ȵı仯�����
�ʴ�Ϊ��
��1��CaCO3+2HCl�TCaCl2+H2O+CO2����
��2���٣��ܣ�
��3��a����
��4���¶�
�����������������ȡ�ǻ�ѧʵ�鿼��Ļ����ͳ�������ʽ���ڻش�ʱһ��Ҫע�������ϵ�Ķ�Ӧ��ͬʱע����������Ҫȷ��λ�����ƿ��ʹ���ǡ�ϴ��ƿ�dz����̳�������ƿ�Ƕ̽���������

��ϰ��ϵ�д�
 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�
�����Ŀ
ͬѧ��ѧϰ�ˡ���������һ�ºԽ������й����ʲ�����Ũ�����Ȥ��
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
��2�����Ӳ�ʲ������ں�ɫ��ĩΪ�������ʵĻ���Ҫȷ����ĩ���������ʵ���������������Ҫͨ���й����ݼ�������жϣ���������Ϊ�������ݵ��ǣ� ��
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
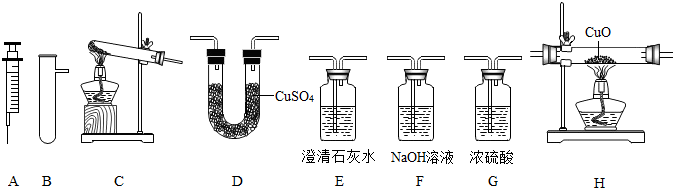
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a b�������������=����
�ڲ������������ʣ�a b�������������=����
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺
����Ʒ��ͭ����������Ϊ�� ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ �ˣ�
̽��һ��ijС����������ͼ��ʾ��̽��ʵ�飮ʵ�鷢��CO��CuO���Ⱥ��ɫ��ĩ��ɺ�ɫ��ĩ����������С����о����̲�����������

[�о�����]̽����ɫ��ĩ����Ҫ�ɷ�
[��������]
��1���й����ʵ���ɫ��CuO��ĩ����ɫ��Cu2O��ĩ����ɫ
��2��CuO��Cu2O���ܺ�ϡ���ᷢ����Ӧ����ѧ����ʽΪ��
CuOʮH2SO4=CuSO4+H2O
Cu2O+H2SO4=CuSO4+Cu+H2O
[������ʵ��]
��1�����Ӳ�ʲ������ں�ɫ��ĩΪһ�����ʣ���������ijɷ֣�����Ƽ�ʵ��֤����IJ²⣮
| ���� | ��ʵ�鷽�� | ���� | CO��CuO��Ӧ�Ļ�ѧ����ʽ |
| ��ĩΪCu | ����Ӧ | CuO+CO=Cu+CO2 | |
| ��ĩ�ܽ⣬��Һ�����ɫ�����ɺ�ɫ��ĩ | 2CuO+CO=Cu2O+CO2 |
A����ӦǰCuO��ĩ��������B��Ӳ�ʲ������й������ʼ��ٵ�����
C��ͨ��CO��������D����Ӧ����������������
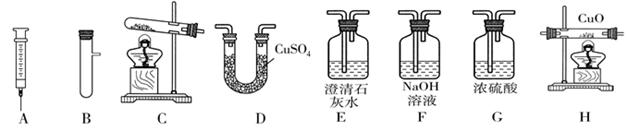
̽������ʵ����ͨ�����÷�Ӧ��Zn+H2SO4=ZnSO4+H2����������������
��1��̽��С�鷢��ʵ�������������ô�п�������ʵ�п�������ô�п��ԭ���Ǵ�п�����ᷴӦ�ų����������ʱȴ�п�죮�ֽ������������ݴ�п�۷ֱ����a��b�����ձ��У����ձ��о���ʢ����ͬ�Ĺ���ϡ���ᣬͬʱ��a�ձ��м���������CuO��ĩ����ַ�Ӧ�����ձ��У�
�ٲ���������������a
�ڲ������������ʣ�a
��2��Ϊ�˲ⶨ��ͭ��ͭ��п�Ͻ𣩵���ɣ�ij�о���ѧϰС���ȡ����Ʒ10g����������μ���9.8%��ϡ�������պò��ٲ�������Ϊֹ����Ӧ���������ɵ�����������������Һ��������ϵ����ͼ��ʾ���Լ��㣺

����Ʒ��ͭ����������Ϊ��
��ǡ����ȫ��Ӧʱ������ϡ���������Ϊ
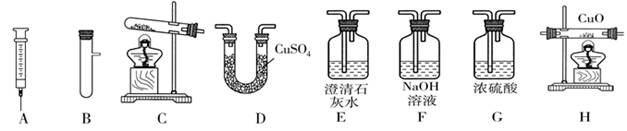
 ij��ȤС����������װ�ý���O2��CO2��ʵ�����Ʒ����й����ʵ��о���
ij��ȤС����������װ�ý���O2��CO2��ʵ�����Ʒ����й����ʵ��о���