��Ŀ����
��8�֣�ijʵ��С�齫����Ƥ����ԭ����ʯ�ҡ������ʳ�η���ˮ�У���ַ�Ӧ����ˣ��õ�������Һ�Ͱ�ɫ���壺
��1�����������з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2���ҵ��Ʋ��ǣ�����Һ��һ�����ڵ������� ��
��3��Ϊ��һ��ȷ������Һ���п��ܵ���ɣ�ͬѧ���������ͼ��ʾ��ʵ��װ�ý�����֤��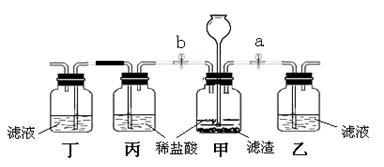
�ټ�װ���з�����Ӧ�Ļ�ѧ����ʽΪ ��
�ڹر�ֹˮ��b����ֹˮ��a�����۲쵽��װ�ó��ֻ�����������Һ�е������� ��
�۹ر�ֹˮ��a����ֹˮ��b��ʵ��Ŀ���� ��Ԥ���ʵ������Ϊ ��
�ܴ�a��b�� ����д����ͽ��ۣ���
��CaO+H2O=Ca(OH)2 Ca(OH)2 + Na2CO3 CaCO3��+ 2NaOH
������2�֣����һ������ʽ��1�֣�
�� NaCl��NaOH
�� �� CaCO3 + 2HCl CaCl2 + H2O + CO2��
��NaCl NaOH Ca(OH)2
����֤������Һ�ﺬ��̼���� ����Һ�������������ݲ���
�ܶ��������ݲ��������в����ǣ�������Һ��ֻ��NaCl��NaOH��
���������������1����ʯ�ҡ������ʳ�η���ˮ�У���ʯ����ˮ��Ӧ��������ʯ�ң���ʯ���봿�Ӧ������̼��Ƴ������������ƣ���Ӧ�Ļ�ѧ����ʽΪ��CaO+H2O=Ca(OH)2��Ca(OH)2 + Na2CO3 CaCO3��+ 2NaOH��
��2���ɣ�1���еķ�Ӧ��֪������Һ��һ�����ڵ�������NaCl��NaOH��
��3���ټ�װ���з�����Ӧ�Ļ�ѧ����ʽΪCaCO3 + 2HCl CaCl2 + H2O + CO2����
�ڹر�ֹˮ��b����ֹˮ��a�����۲쵽��װ�ó��ֻ�����������Һ�е�������NaCl��NaOH��Ca(OH)2��
�۹ر�ֹˮ��a����ֹˮ��b��ʵ��Ŀ������֤������Һ�ﺬ��̼���ƣ�Ԥ���ʵ������Ϊ����Һ�������������ݲ�����
�ܴ�a��b�������������ݲ��������в����ǣ�����Һ��ֻ��NaCl��NaOH��
���㣺�����ƶϣ�ʵ�������̽������ѧ����ʽ��ʵ��װ�õ�ʹ�á�
���������еĻ�ѧ��Ӧ�������ƶ����ʵ��������ݣ��������еĻ�ѧ��Ӧ�������ƶ����ʣ��dz��õ��ƶϷ�����









