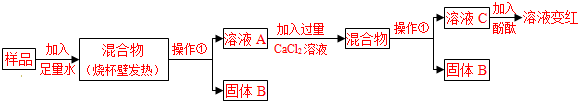
��Ŀ����
�ټ�ʯ����ʵ���ҳ��õĸ���������������ơ��������ƵĹ������ ����������ˮ�����⣬��ʯ�һ������ճ����������� ��9�� ��ѡ���ţ�ֻ��һ����������ѡ���÷֣���
A������ B��һ����̼ C��������̼ D������
��ʵ���������ò����������壨FeC2O4��2H2O�����ȷֽ�õ�һ������CO����Ӧ�ķ���ʽ Ϊ��FeC2O4��2H2O�� ����������+CO+CO2+2H2O

 �÷�Ӧ���� ��10�� ����д������Ӧ���ͣ��� ���������غ㶨�ɣ�������֪�����С�����������Ļ�ѧʽ�� ��11����
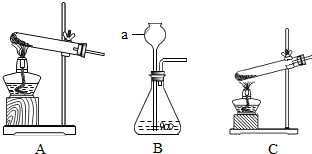
�÷�Ӧ���� ��10�� ����д������Ӧ���ͣ��� ���������غ㶨�ɣ�������֪�����С�����������Ļ�ѧʽ�� ��11�����ۼ��Ȳ���������ѡ�����С�A��B��C������װ���е���12�� ��ѡ���š�A��B��C������
|
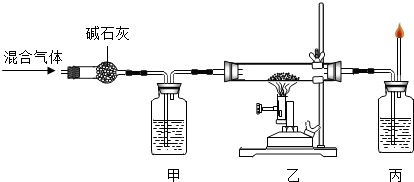
�ܲ������������Ħ����������14����дȫ��λ��������1.8g�������������ַ�Ӧ�ɵ� ��CO��������ʵ����Ƕ��٣�
��д��������̣���15��
|
д�����ҡ�����Ӧ�������� ��16�� ����Ӧ�Ļ�ѧ����ʽΪ
 ��17�� ���÷�Ӧ�л�ԭ���� ��18�� �� �ס���������ʢ�ŵ���Һ��
��17�� ���÷�Ӧ�л�ԭ���� ��18�� �� �ס���������ʢ�ŵ���Һ������ ��19�� ����20�� �� ���С��ס�����ʢ�Ÿ���Һ
�������� ��21�� ��
��9��C ��10���ֽⷴӦ ��11��FeO ��12��A ��13������©��
��14��180��/Ħ�� ��15��0.01mol
��16����ɫ������� ��17��2FeC2O4==Fe2O3+3CO+CO2
(18) CO (19)������Һ ��20��������Һ
��21�����鲢��ȥ��������еģ�����Ӱ��ʵ����ۡ�����:
��
��14��180��/Ħ�� ��15��0.01mol
��16����ɫ������� ��17��2FeC2O4==Fe2O3+3CO+CO2
(18) CO (19)������Һ ��20��������Һ
��21�����鲢��ȥ��������еģ�����Ӱ��ʵ����ۡ�����:
��

��ϰ��ϵ�д�
�����Ŀ


 33����ʯ����ʵ���ҳ��õĸ������
33����ʯ����ʵ���ҳ��õĸ������
 ��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����
��ʯ����ʵ���ҳ��õĸ������ͬѧ��Ϊȷ��һƿ���õġ���ʯ�ҡ��������Ѳ��ֱ��ʻ�ȫ�����ʣ���Ʒ�ijɷ֣���������̽����