��Ŀ����
Ϊ�ⶨ���Ȼ��ƺ��Ȼ�����ɵĹ�����Ʒ���Ȼ��Ƶĺ�����ijͬѧ����������ʵ�飺ȡ14�˹�����Ʒ��ȫ������100��ˮ�У������õĻ����Һ�еμ�������������Ϊ10.6%0��̼������Һ����¼����������ͼ��ʾ�����ߣ�
��1�����Ȼ�����̼����ǡ����ȫ��Ӧʱ�����ó���������Ϊ______�ˣ���ʱ��Ҫ�õ��������Ȼ��ƹ��壬Ӧ���еIJ�����______��
��2����Ʒ���Ȼ��Ƶ����������Ƕ��٣�
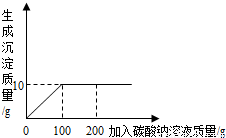
���𰸡���������1������ͼ�õ������������������Ȼ��Ƶ��ܽ�ȿ��ǽᾧ��������2�����ݳ�������������Ȼ��Ƶ��������ٳ���14g���У�
����⣺��1����ͼʾ��֪����10g�����������ӣ�˵����Ӧ���������10g����������ܽ�����¶�Ӱ��ϴ�����ʴ���Һ�нᾧ�����ķ�������ȴ�ȱ�����Һ������ܽ�����¶�Ӱ�첻������ʴ���Һ�нᾧ�����ķ����������ܼ��������Ȼ��Ƶ��ܽ�����¶�Ӱ���С�������������ܼ��������Ƚ��������˳�ȥ��������
��2���⣺����Ʒ���Ȼ��Ƶ�����Ϊx
CaCl2+Na2C03=CaC03��+2NaCl
111 100
x 10��
���ݣ�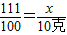 ���x=11.1��
���x=11.1��
����Ʒ���Ȼ��Ƶ���������Ϊ��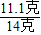 ×100%=79.3%��
×100%=79.3%��
�ʴ�Ϊ����1��10���Ƚ��պó�����ȫʱ���õĻ������ˣ��ٽ��õ�����Һ��������ɣ���2��79.3%��
��������������׳����ĵط����Ȼ��ƽᾧ�����ķ������������Ƚ��������˵���������
����⣺��1����ͼʾ��֪����10g�����������ӣ�˵����Ӧ���������10g����������ܽ�����¶�Ӱ��ϴ�����ʴ���Һ�нᾧ�����ķ�������ȴ�ȱ�����Һ������ܽ�����¶�Ӱ�첻������ʴ���Һ�нᾧ�����ķ����������ܼ��������Ȼ��Ƶ��ܽ�����¶�Ӱ���С�������������ܼ��������Ƚ��������˳�ȥ��������
��2���⣺����Ʒ���Ȼ��Ƶ�����Ϊx
CaCl2+Na2C03=CaC03��+2NaCl
111 100
x 10��
���ݣ�
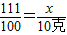 ���x=11.1��
���x=11.1������Ʒ���Ȼ��Ƶ���������Ϊ��
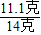 ×100%=79.3%��
×100%=79.3%���ʴ�Ϊ����1��10���Ƚ��պó�����ȫʱ���õĻ������ˣ��ٽ��õ�����Һ��������ɣ���2��79.3%��
��������������׳����ĵط����Ȼ��ƽᾧ�����ķ������������Ƚ��������˵���������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ

 С�������������������ʸ����ȴ�����º�Ƶù�������Ϊ13.1g����Ʒ��̼���Ƶ���������Ϊ
С�������������������ʸ����ȴ�����º�Ƶù�������Ϊ13.1g����Ʒ��̼���Ƶ���������Ϊ



 С�������������������ʸ����ȴ�����º�Ƶù�������Ϊ13.1g����Ʒ��̼���Ƶ���������Ϊ______������Ҫ��д��������̣������·�д����
С�������������������ʸ����ȴ�����º�Ƶù�������Ϊ13.1g����Ʒ��̼���Ƶ���������Ϊ______������Ҫ��д��������̣������·�д����