��Ŀ����
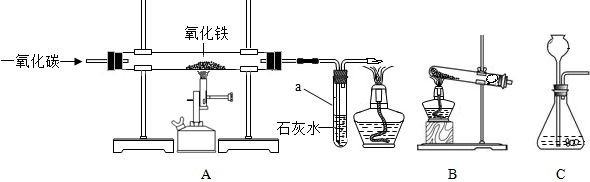
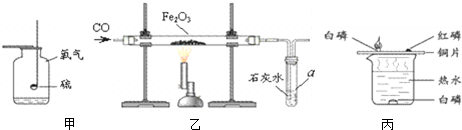
�������ͼ��ʾʵ�飬�ش����⣮

��1�����пɹ۲쵽����������ȼ�գ�����
��2����������a��������
��3������ͭƬ�ϵ�������

��1�����пɹ۲쵽����������ȼ�գ�����
����������ɫ
����������ɫ
���棬�˷�Ӧ�Ļ�ѧ����ʽ��S+O2
SO2
| ||
S+O2
SO2
��
| ||
��2����������a��������
�Թ�
�Թ�
���������е���������ɫ��ĩ���
��ɫ��ĩ���
����װ�õIJ���֮����û��β������װ��
û��β������װ��
����3������ͭƬ�ϵ�������
����ȼ�գ������������̣����ײ�ȼ��
����ȼ�գ������������̣����ײ�ȼ��
���ɴ˵ó���ȼ��ȼ�յ��������¶���Ҫ�ﵽ�ÿ�ȼ����Ż��
�¶���Ҫ�ﵽ�ÿ�ȼ����Ż��
����������1��Ҫע���������ڿ�����������ȼ�յ��������𣬷���ʽ��дҪ��Ҫ��������ƽ��
��2����Ϥ�������ƣ����ݷ�Ӧǰ�����ʵı仯�������������CO�ж���ʵ���ұ���ע��β��������
��3������ȼ�յ��������з�����
��2����Ϥ�������ƣ����ݷ�Ӧǰ�����ʵı仯�������������CO�ж���ʵ���ұ���ע��β��������
��3������ȼ�յ��������з�����
����⣺
��1�����ڿ���ȼ�գ�S+O2
SO2���е���ɫ���棬��������ȼ�գ�������������ɫ���森
�ʴ�Ϊ������������ɫ��S+O2
SO2
��2��a����Ϊ���Թܣ���������Ӧ���Ϊ����������ɫ�ɺ�ɫ��Ϊ��ɫ����ΪCO�ж�����Ҳ�������ʯ��ˮ��Ӧ������ȱ��β��������
�ʴ�Ϊ���Թܣ���ɫ��ĩ��ڣ�û��β������װ��
��3�����ڰ����Ż��ͣ�������ȼ�գ������ײ�ȼ�գ�����ȼ�ղ��������İ��̣�ͨ���Աȿ�ȼ��Ҫȼ�ճ��������Ӵ��⣬������õ��Ż�㣮
�ʴ�Ϊ������ȼ�գ������������̣����ײ�ȼ�գ��¶���Ҫ�ﵽ�ÿ�ȼ����Ż��
��1�����ڿ���ȼ�գ�S+O2
| ||
�ʴ�Ϊ������������ɫ��S+O2
| ||
��2��a����Ϊ���Թܣ���������Ӧ���Ϊ����������ɫ�ɺ�ɫ��Ϊ��ɫ����ΪCO�ж�����Ҳ�������ʯ��ˮ��Ӧ������ȱ��β��������
�ʴ�Ϊ���Թܣ���ɫ��ĩ��ڣ�û��β������װ��
��3�����ڰ����Ż��ͣ�������ȼ�գ������ײ�ȼ�գ�����ȼ�ղ��������İ��̣�ͨ���Աȿ�ȼ��Ҫȼ�ճ��������Ӵ��⣬������õ��Ż�㣮
�ʴ�Ϊ������ȼ�գ������������̣����ײ�ȼ�գ��¶���Ҫ�ﵽ�ÿ�ȼ����Ż��
������������Ҫ������ѧ���Կα�����֪ʶ�����գ�ѧ����ѧϰ��Ҫע��α�����֪ʶ�ĸ�ϰ�����������⣬Ҫ��ѧϰ������ѧ����α�Ҫ�𰸣�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ