��Ŀ����
3����1��θ��ƽ����Ҫ�ɷ�����������������������θ����࣬��ԭ���ǣ��û�ѧ����ʽ��ʾ��Al��OH��3+3HCl�TAlCl3+3H2O����2��ˮ��һ�ֺܺõ��ܼ������м��ּ�������������ˮ���ܼ����ƶ��ɵģ��书�ܼ���Ч�ɷ����±���ʾ��������±��ش��������⣺
| �������� | ����� | ������Ư | Ư�� |
| ���� | ��Ч����۹�������ζ | Ưϴ���ʹɫ�ʸ����� | ����Ʈ���������� |
| ��Ч�ɷ� | HCl | H2O2 | NaClO |
�ڡ�����顱�롰Ư�������ܻ��ã�����������ײ����ж���������ͬʱ���Ȼ��ƺ�ˮ��������÷�Ӧ�Ļ�ѧ����ʽΪ2HCl+NaClO=NaCl+H2O+Cl2����
��3�������������ϵ�̼����͵�̼����������Ǣ٢ۢܣ�����ĸ��ţ���
�ٷ��ദ������������ �ڶ�ʹ�����ϴ���������Ʒ
�ۣ�Ϊ�˽�Լ��ֽ������ʹ��ֽ�� ���������ֹرյ�����Դ��ϰ�ߣ�
���� ��1������θ���к������ᣬ�����ܺ�����������Ӧ�����Ȼ�����ˮ���
��2��
�ٹ��������ڶ������̵Ĵ�������������������
�ڸ��ݷ�Ӧ��������P�������غ㶨�ɿ�����д��ѧ����ʽ��
��3����̼����͵�̼����Ҫ�������������������ĵ��������Ӷ�����̼���ŷţ����Դӽڵ硢���ܺͻ��յȻ������ı��������ϸ�ڣ����з�����ɣ�
��� �⣺
��1���������������ᷴӦ�Ļ�ѧ����ʽΪ��Al��OH��3+3HCl�TAlCl3+3H2O��
��2��
�ٹ��������ڶ������̵Ĵ���������������������������ݲ�����
�ڡ�����顱�롰Ư��������ܷ�����ѧ��Ӧ����Ӧ�Ļ�ѧ����ʽΪ��2HCl+NaClO=NaCl+H2O+Cl2����
��3��
�ٷ��ദ���������������ܽ�Լ��Դ�����ٻ�����Ⱦ����ѡ����ϵ�̼����͵�̼�������
�����ϴ���������Ʒ��������������Դ����Դ����ʹ�����ϴ���������Ʒ�����������ĵ�������������̼���ŷš���ɫ��Ⱦ�ȣ���ѡ����ϵ�̼����͵�̼�������
��Ϊ�˽�Լ��ֽ������ʹ��ֽ�ţ��ܼ���ľ�ĵ�ʹ�ã��Ӷ����ٶ�����̼���ŷţ���ѡ����ϵ�̼����͵�̼�������
���������ֹرյ�����Դ��ϰ�ߣ��ܽ�Լ���ܣ����ٶ�����̼���ŷţ���ѡ����ϡ���̼������
�𰸣�
��1��Al��OH��3+3HCl�TAlCl3+3H2O
��2���������ݲ��� ��2HCl+NaClO=NaCl+H2O+Cl2����
��3���٢ۢ�
���� �����������еij�������Ϊ����㣬����������֮��ķ�Ӧ���Լ���ѧ����ʽ��������⣮����ѧ����ѧϰ��ѧ����Ȥ��

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ���ˮ������ȼ�յĻ�����Ӧ������ͬ | |
| B�� | ��Ȫˮ������Ԫ�غ���Ԫ����ɵ������� | |
| C�� | ����ˮ�ij��÷��������������������˺����� | |
| D�� | ��Ӳˮ���÷���ϴ�·��������ĭ�ม��������� |
| A�� | þ���ڿ�����ȼ�գ������⣬ð��Ũ�ҵĺ��̣��ų����������ɺ�ɫ��ĩ | |
| B�� | ��˿�������о���ȼ�գ������Ľ����ų����������ɺ�ɫ��ĩ | |
| C�� | ľ̿�ڿ�����ȼ�գ�������⣬�ų�������������ʹ����ʯ��ˮ����ǵ����� | |
| D�� | ����������ȼ�գ��������ĵ���ɫ���棬�ų�����������û����ζ������ |
| A�� | �÷�Ӧ�����û���Ӧ | B�� | ��Ӧ��M�Ļ��ϼ�Ϊ+2�� | ||
| C�� | M�Ľ�����Ա�Agǿ | D�� | ����M�������� |
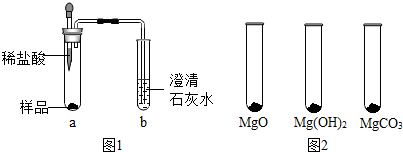
��֪��MgO+2HCl=MgCl2+H2O MgCO3+2HCl=MgCl2+H2O+CO2��
��1��̽����þ�ۡ����Ƿ���Mg��MgCO3��
��ȡ������Ʒ����ͼ1��ʾ����ʵ�飬�۲쵽a�Թ��������ݲ�����b�Թ��в����������dz���ʯ��ˮ����ǣ���֤����þ�ۡ���һ������MgCO3��
��Ϊ֤����þ�ۡ����Ƿ���Mg��С����ȼ�ŵ�ľ������ͼ��b�Թܿ��Ϸ���ľ��Ϩ�𣬵�ͬѧ��ָ������ʵ�鲻�ܴ��ʵ��Ŀ�ģ���Ҫ��ͼ�еij���ʯ��ˮ�滻��ŨNaOH��Һ��Ŀ���ǰѶ�����̼ȫ�����գ�С�������ĺ��ʵ�鷽���ظ�����ʵ�飬�۲쵽b�Թܿ�����������֤����þ�ۡ���û��Mg��
��2��̽����þ�ۡ����Ƿ���MgO��Mg��OH��2��
��ʵ����̡�
����MgO��Mg��OH��2��MgCO3���ֹ���ֱ�������ʵ�飬�ֱ�ȡ0.5g���ֹ����ĩ��ÿ��ȡ��������ͼ2��ʾ��
��μ�����ͬ��������������ϡ����ֱ����ĩǡ����ʧ�����±��м�¼���ĵ�ͬŨ��ϡ���������������������ͬһ�����²ⶨ���ұ�����С�����1λ��
| MgO | Mg��OH��2 | MgCO3 | |
| ����ϡ��������/ml | 10.4 | 7.2 | 5.0 |
��ȡ��þ�ۡ���Ʒ0.5g����������ϡ��������ĩǡ���ܽ⣬��ʱ����ϡ��������ԼΪ5.3mL��
��ʵ����������ۡ�
��þ�ۡ���ֻ����MgCO3��������0.5g��þ�ۡ�����ϡ����������0.5gMgCO3����ϡ������������ȣ�
��ʵ�鷴˼��
Ϊȷ����þ�ۡ��ľ���ɷ֣�ͬѧ����Ϊ����Ҫ��������ʵ�飺�ֱ�ȡ0.5g��þ�ۡ���0.5gMgCO3��������ϡ���ᣬ�ⶨ���ɵ���������ֱ�Ϊ119ml��140ml���ɴ˿�֪��þ�ۡ��к���MgCO3����������Ϊ85%��
| A�� | ϡ�����еμ���ɫʯ����Һ����Һ���� | |
| B�� | ϸ��˿��������ȼ�ղ�������ɫ���棬���ɰ�ɫ���� | |
| C�� | �����������Һ�м��������������̣���Ӧֹͣ���ɫ������ʧ | |
| D�� | ��̼������Һ�м������ʯ��ˮ���а�ɫ�������� |