��Ŀ����
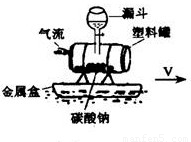
��ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�Һ����̼���Ʒ�Ӧ��������������̼���壬���Ϲ�����ѹѸ����������ӹ�С�������ʹ��ǰ������ش��������⣺��1��©���е�Һ������ǣ�д��һ�֣� ��
��2����ֹ��ˮ������������������������ǰ������һ����˵���� ��
��3����������ǰ��������ˮ�ĸ�����α仯�� ��
���𰸡���������1��ǿ����η������ֽⷴӦ��
��2����������Ч���������������Ըı�������˶�״̬�������Ըı��������״��
��3����Ϊ������Ư��״̬���ܵĸ����������ܵ�������F��=G��
����⣺��1�����ܺ�̼���η������ֽⷴӦ������������̼���壮��Ӧ����ʽΪ��H2SO4+Na2CO3�TNa2SO4+H2O+CO2��
��2�������������壬������һ������������Ϊ������������ģ���ô�������ͻ����һ���������������ڷ��������������£��ı���״̬���ɾ�ֹ��Ϊ�˶�����������ı���������˶�״̬��
��3��������Ư��״̬�������ܵĸ���������������F��=G����Ϊ�����Ϲ��з�����ѧ��Ӧ��������������������������ر�С�����Դ��ܵĸ���Ҳ���С��
�ʴ�Ϊ����1��ϡ�����2��������������ģ���3����С��
������������һ���ۺ��Ժ�ǿ����Ŀ���漰����֪ʶ�ܶ࣬�л�ѧ��Ӧ����������Ч����Ư��ʱ���ܸ����Ĵ�С����Ҫѧ���к�ǿ������֪ʶ��������
��2����������Ч���������������Ըı�������˶�״̬�������Ըı��������״��
��3����Ϊ������Ư��״̬���ܵĸ����������ܵ�������F��=G��
����⣺��1�����ܺ�̼���η������ֽⷴӦ������������̼���壮��Ӧ����ʽΪ��H2SO4+Na2CO3�TNa2SO4+H2O+CO2��
��2�������������壬������һ������������Ϊ������������ģ���ô�������ͻ����һ���������������ڷ��������������£��ı���״̬���ɾ�ֹ��Ϊ�˶�����������ı���������˶�״̬��
��3��������Ư��״̬�������ܵĸ���������������F��=G����Ϊ�����Ϲ��з�����ѧ��Ӧ��������������������������ر�С�����Դ��ܵĸ���Ҳ���С��
�ʴ�Ϊ����1��ϡ�����2��������������ģ���3����С��
������������һ���ۺ��Ժ�ǿ����Ŀ���漰����֪ʶ�ܶ࣬�л�ѧ��Ӧ����������Ч����Ư��ʱ���ܸ����Ĵ�С����Ҫѧ���к�ǿ������֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ

 ��2010?����ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮
��2010?����ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮ ��ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮
��ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮
 ��ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮
��ͼ��ʾ��ij�Ƽ�С����Ʋ�������һֻ����������������������Ӿ�ص�ˮ���ϣ���©���Ļ��������µ�������̼���Ʒ�Ӧ��������������̼���壬���Ϲ�������Ѹ����������ӹ�С�������ʹ��ǰ������ش��������⣮