��Ŀ����
�ҹ��Ƽҵ��������°��Ƽ����ư���������������Ƽ��Ϊ����͵��ʹ�ҵ�����ķ�չ�����˽ܳ��Ĺ��ס���������������ʾ��ͼ���£�
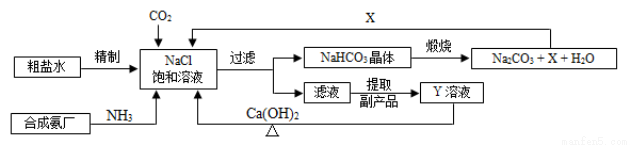
��1������ˮ����Ҫ����Ca2����Mg2����SO42�����������ӣ���ҵ�ϳ��ü����Թ����Ģ� NaOH��Һ���� Na2CO3��Һ���� BaCl2��Һ������������ȳ��ӡ����ơ�������Լ�������˳��Ϊ_____________������ĸ��ţ���
A���٢ڢ� B���ۢڢ� C���٢ۢ�
��2��������������ǣ��÷���ʽ��ʾ��________________��______________��
��3���Ƽ�ԭ���ɿ������ɢ� CO2 + NH3 + H2O = NH4HCO3�͢� NaCl + NH4HCO3 = NaHCO3+ NH4Cl������Ӧ�Ӻ϶��ɡ���Ӧ��Ϊ���ֽⷴӦ������ݸ��ֽⷴӦ��������������1���������20��ʱ���ܽ�ȣ�������Ӧ��Ϊ����˳�����У�_______________��
��1 ������ʵ��ܽ�ȣ�20�棩����λ��g��
NaCl | NH4HCO3 | NaHCO3 | NH4Cl |
36.0 | 21.7 | 9.6 | 37.2 |
��4������2Ϊϴ�ӡ����ա�����Ӧ������װ��______________��(����ĸ��ţ����С�ϴ�ӡ�������

�����������У�����жϾ����Ѿ�ϴ�Ӹɾ�______________��
��5��X�Ļ�ѧʽΪ_____________����Xֱ���ŷŵ������л���ɵĺ����_______________��
��6��Y����Һ������Ҫ�ɷ֣�Y��Ca(OH)2��Ӧ�IJ���֮һ������ѭ��ʹ�ã��ò��������Ϊ______________��Y�Ļ�ѧʽΪ______________��д��Y��һ����;_____________��
 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�

 2Fe+3CO2
2Fe+3CO2  2Na+Cl2��
2Na+Cl2��