��Ŀ����
Ϊ�ⶨij̼����炙����еĺ�������С����ʵ������ȡ��8.5 g�û�����Ʒ��20 g NaOH��Һ���ȣ�ǡ����ȫ��Ӧ����ò�����Ϊ26.8 g(��ʾ����Ӧ�Ļ�ѧ����ʽΪNH4HCO3��2NaOH=Na2CO3��2H2O��NH3�������ɵİ���ȫ���ݳ��������ɷֲ�������Ҳ���μӷ�Ӧ)������
(1)���ɰ�����������
(2)�û����е�Ԫ�ص���������(��������ȷ��0.1%)��
(3)������Һ����������������
���𰸡�
(1)1.7 g (2)16.5% (3)40%
��������(1)���������غ㶨�ɣ���������ɰ���������Ϊ8.5 g��20 g��26.8 g��1.7 g��
(2)��μӷ�Ӧ��̼����淋�����Ϊx���μӷ�Ӧ���������Ƶ�����Ϊy
NH4HCO3��2NaOH=Na2CO3��2H2O��NH3��
79?????? 80????????????????? 17
x??????? y?????????????????? 1.7 g
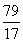 ��
��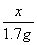
x��7.9 g
̼����狀���Ԫ�ص�����Ϊ7.9 g��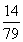 ��1.4 g
��1.4 g
̼����炙����е�Ԫ�ص���������Ϊ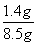 ��100%��16.5%
��100%��16.5%
(3) 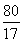 ��
��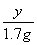
y��8 g
NaOH��Һ��������������Ϊ��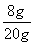 ��100%��40%
��100%��40%

��ϰ��ϵ�д�
�����Ŀ