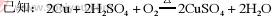��Ŀ����
ͭ�������������õĽ���֮һ��
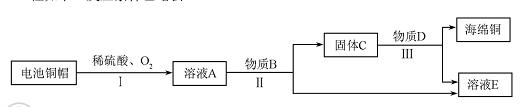
��1������ͭ��Ʒ�У����ý��������Ե���������������ĸ��ţ���

A��ͭ�ʽ��� B��ͭ���� C��ͭ���
��2����ʪ����ͭ����ԭ��������ͭ��Һ������Ӧ���÷�Ӧ����������Ӧ����Ϊ��������
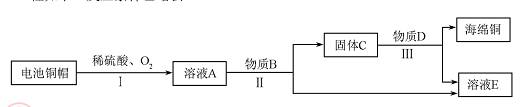
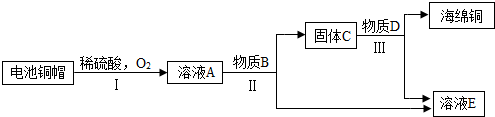
 ��3�����÷Ͼɵ��ͭñ���� Cu��Zn����ȡ����ͭ��Cu�������õ�����п��Һ����Ҫ�������£���Ӧ��������ȥ����
��3�����÷Ͼɵ��ͭñ���� Cu��Zn����ȡ����ͭ��Cu�������õ�����п��Һ����Ҫ�������£���Ӧ��������ȥ����
|
��֪��2Cu+2H2SO4+O2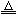 2CuSO4+2H2O
2CuSO4+2H2O
�ٹ��̢��з����������������������
�ڹ��̢���������������÷�Ӧ�Ļ�ѧ����ʽΪ ������ ��
��A~D�к�ͭ��п����Ԫ�ص�������������������ĸ��ţ���
��5�֣���1��C ��2���û���Ӧ
��3���ٹ��� ��Zn+H2SO4= ZnSO4+H2�� ��AC

��ϰ��ϵ�д�
 �¿α�ͬ��ѵ��ϵ�д�
�¿α�ͬ��ѵ��ϵ�д� һ����ʦ����Ӧ����������һ��ȫϵ�д�
һ����ʦ����Ӧ����������һ��ȫϵ�д� Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д�
�����Ŀ
 ͭ�ʽ��� B��
ͭ�ʽ��� B�� ͭ���� C��
ͭ���� C�� ͭ���
ͭ���

 2CuSO4+2H2O
2CuSO4+2H2O 

 2CuSO4+2H2O
2CuSO4+2H2O