��Ŀ����
����С���ͬѧ�ڲⶨ��NaCl ��Na2CO3�γɵĹ����������ʱ������������ʵ�飺ȡ40�˹������������Һ��ƽ����Ϊ4�ݣ�Ȼ��ֱ����һ������������CaCl2��Һ��ʵ�����±���
| ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
| ԭ������������� | 10g | 10g | 10g | 10g |
| ����CaCl2��Һ���� | 10g | 20g | 0g | 40g |
| ���ɵij��������� | 2g | m | 5g | 5g |
��1��m=______�����ɵij����ǣ���д��ѧʽ��______��
��2��10gԭ����������ɵ���Һ������CaCl2��Һ��Ӧ��������ɳ�������Ϊ______g��
��3��ԭ����������Na2CO3�����������Ƕ��٣�
�⣺��1���ڷ�Ӧ�����У��μӷ�Ӧ������������������������ȣ���ͼ����֪��ÿ10gCaCl2��Һ������2g��������m=4g���������֪�˷�Ӧ�ķ���ʽΪCaCl2+Na2CO3=CaCO3��+2NaCl���ʳ�������CaCO3��
��2����ͼ����֪������CaCl2��Һ��������30g��40gʱ�ij�������һ���࣬˵���ӵ�30g ʱ����ȫ��Ӧ����������ɳ�������Ϊ5g��
��3����ͼ����֪�������е�Na2CO3��෴Ӧ����5g��������10gԭ����������Na2CO3����Ϊx
CaCl2+Na2CO3=CaCO3��+2NaCl
106 100
x 5g
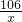 =
=
x=5.3g
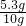 ��100%=53%
��100%=53%
��ԭ����������Na2CO3������������53%��
��������1����֪m��ֵ����֪��Ӧ�����и�����֮��Ĺ�ϵ��Ҫ֪���ɵij����Ǻ����ʣ�����ݲμӷ�Ӧ�����ʵĻ�ѧ����ʽ�����ж���
��2���������ij�����������Ӧ����ȫ��Ӧ�����ɵij�������������������ɳ����������ɷ���ͼ���õ���
��3�����ݷ�Ӧ�Ļ�ѧ����ʽ�����Եõ�������֮��ı�����ϵ���������̼���Ƶ��������ٸ���ԭ����������Na2CO3����������= ��100%���������ԭ����������Na2CO3������������
��100%���������ԭ����������Na2CO3������������
������������Ҫ����ѧ�����ݻ�ѧ����ʽ�ļ��㼰�������ݽ�������������Ҫ������ϸ�ķ������
��2����ͼ����֪������CaCl2��Һ��������30g��40gʱ�ij�������һ���࣬˵���ӵ�30g ʱ����ȫ��Ӧ����������ɳ�������Ϊ5g��
��3����ͼ����֪�������е�Na2CO3��෴Ӧ����5g��������10gԭ����������Na2CO3����Ϊx
CaCl2+Na2CO3=CaCO3��+2NaCl
106 100
x 5g
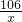 =
=
x=5.3g
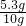 ��100%=53%
��100%=53%��ԭ����������Na2CO3������������53%��
��������1����֪m��ֵ����֪��Ӧ�����и�����֮��Ĺ�ϵ��Ҫ֪���ɵij����Ǻ����ʣ�����ݲμӷ�Ӧ�����ʵĻ�ѧ����ʽ�����ж���
��2���������ij�����������Ӧ����ȫ��Ӧ�����ɵij�������������������ɳ����������ɷ���ͼ���õ���
��3�����ݷ�Ӧ�Ļ�ѧ����ʽ�����Եõ�������֮��ı�����ϵ���������̼���Ƶ��������ٸ���ԭ����������Na2CO3����������=
 ��100%���������ԭ����������Na2CO3������������
��100%���������ԭ����������Na2CO3������������������������Ҫ����ѧ�����ݻ�ѧ����ʽ�ļ��㼰�������ݽ�������������Ҫ������ϸ�ķ������

��ϰ��ϵ�д�
 �Ǽ�����������ϵ�д�
�Ǽ�����������ϵ�д� â���̸������Ծ�ϵ�д�
â���̸������Ծ�ϵ�д�
�����Ŀ
����С���ͬѧ�ڲⶨ��NaCl��Na2CO3�γɵĹ����������ʱ������������ʵ�飺ȡ40g�������������Һ��ƽ����Ϊ�ķݣ�Ȼ��ֱ����һ������������CaCl2��Һ��ʵ�����ݼ��±���
������������ݻش��㣮
��1��m= g��
��2��ԭ����������Na2CO3�����������Ƕ��٣� ��
| ʵ��һ | ʵ��� | ʵ���� | ʵ���� | |
| ԭ������������ | 10g | 10g | 10g | 10g |
| ����CaCl2��Һ���� | 10g | 20g | 30g | 40g |
| ���ɵij��������� | 2g | m | 5g | 5g |
��1��m=
��2��ԭ����������Na2CO3�����������Ƕ��٣�
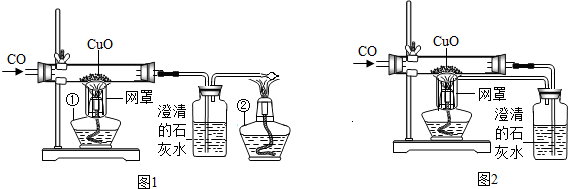
 ijУ����С���ͬѧ����ʦ�İ����£�̽���ó�������Ҫ�ɷ�Fe2O3����������Ҫ��Ӧԭ����������Ƶ�ʵ��װ�ã���ͼ��
ijУ����С���ͬѧ����ʦ�İ����£�̽���ó�������Ҫ�ɷ�Fe2O3����������Ҫ��Ӧԭ����������Ƶ�ʵ��װ�ã���ͼ��