��Ŀ����
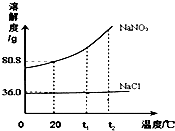 NaNO2��NaCl���ܽ��������ͼ��ʾ���ش��������⣺
NaNO2��NaCl���ܽ��������ͼ��ʾ���ش��������⣺
��1��t1��ʱ��______���ܽ�ȸ���
��2��ͬ���£�______ ����ܹ������ܡ������Ƶ���ͬ�������������ı���NaNO2��NaCl��Һ��
��3������һ��125g��������������Ϊ20%��NaNO2��Һ������Һ�������ʵ�����Ϊ______����ά���¶���20�棬Ҫʹ����Һ��Ϊ������Һ����Ҫ���룮NaNO2______ g��
��4������һ���������Ȼ��Ƶ�NaNO2���壬Ϊ��ȡ������NaNO2�����õķ�����______��
�⣺��1��t1��ʱ��NaNO2���ܽ�ȸ���
��2����ͼ��֪��ͬ���¶��ߵ��ܽ��û����ͬ��ʱ��ϱ�����Һ�����ʵ������������㷽����֪��ͬ���£������Ƶ���ͬ�������������ı���NaNO2��NaCl��Һ��
��3������һ��125g��������������Ϊ20%��NaNO2��Һ������Һ�������ʵ�����Ϊ125g��20%=25g�� ��ά���¶���20�棬���¶���NaNO2���ܽ����80.8g������Ҫʹ����Һ��Ϊ������Һ����Ҫ����NaNO2������Ϊ80.8g-25g=55.8 g��
��4����ͼ��֪�Ȼ��Ƶ��ܽ�����¶�Ӱ���С���������Ƶ��ܽ�����¶�Ӱ��ϴ����Ժ������Ȼ��Ƶ�NaNO2���壬Ϊ��ȡ������NaNO2���ɲ��ý��½ᾧ����ȴ�ȱ�����Һ�ķ�����
�ʴ�Ϊ����1��NaNO2����2�����ܣ���3��25g��55.8����4�����½ᾧ����ȴ�ȱ�����Һ��
��������1�����ܽ�����߿ɱȽ�ͬһ�¶������ʵ��ܽ�ȴ�С��
��2������ʱ���������ļ���ʽ ��100%����֪�ܽ�����������������ȣ��ݴ˽�������ʵ��ܽ�����߷������
��100%����֪�ܽ�����������������ȣ��ݴ˽�������ʵ��ܽ�����߷������
��3�����ʵ�����=��Һ�����������ʵ���������������ϸ��¶���NaNO2���ܽ�ȷ������
��4�����Ȼ��ƺ��������Ƶ��ܽ�����¶�Ӱ������������
�����������Ĺؼ�����ȷ�ܽ�ȵĸ�������ü���������ʵ������������ܽ�����ߵ����岢�ܽ���������������
��2����ͼ��֪��ͬ���¶��ߵ��ܽ��û����ͬ��ʱ��ϱ�����Һ�����ʵ������������㷽����֪��ͬ���£������Ƶ���ͬ�������������ı���NaNO2��NaCl��Һ��
��3������һ��125g��������������Ϊ20%��NaNO2��Һ������Һ�������ʵ�����Ϊ125g��20%=25g�� ��ά���¶���20�棬���¶���NaNO2���ܽ����80.8g������Ҫʹ����Һ��Ϊ������Һ����Ҫ����NaNO2������Ϊ80.8g-25g=55.8 g��
��4����ͼ��֪�Ȼ��Ƶ��ܽ�����¶�Ӱ���С���������Ƶ��ܽ�����¶�Ӱ��ϴ����Ժ������Ȼ��Ƶ�NaNO2���壬Ϊ��ȡ������NaNO2���ɲ��ý��½ᾧ����ȴ�ȱ�����Һ�ķ�����
�ʴ�Ϊ����1��NaNO2����2�����ܣ���3��25g��55.8����4�����½ᾧ����ȴ�ȱ�����Һ��
��������1�����ܽ�����߿ɱȽ�ͬһ�¶������ʵ��ܽ�ȴ�С��
��2������ʱ���������ļ���ʽ
 ��100%����֪�ܽ�����������������ȣ��ݴ˽�������ʵ��ܽ�����߷������
��100%����֪�ܽ�����������������ȣ��ݴ˽�������ʵ��ܽ�����߷��������3�����ʵ�����=��Һ�����������ʵ���������������ϸ��¶���NaNO2���ܽ�ȷ������
��4�����Ȼ��ƺ��������Ƶ��ܽ�����¶�Ӱ������������
�����������Ĺؼ�����ȷ�ܽ�ȵĸ�������ü���������ʵ������������ܽ�����ߵ����岢�ܽ���������������

��ϰ��ϵ�д�
�����Ŀ
 25����Һ�������Ϳ����о��й㷺��Ӧ�ã������ǵ�����ϢϢ��أ�����������ش����⣮
25����Һ�������Ϳ����о��й㷺��Ӧ�ã������ǵ�����ϢϢ��أ�����������ش����⣮
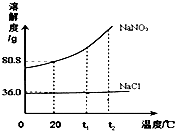 NaNO2��NaCl���ܽ��������ͼ��ʾ���ش��������⣺
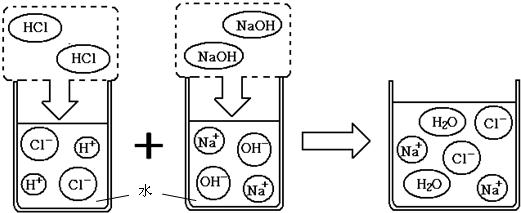
NaNO2��NaCl���ܽ��������ͼ��ʾ���ش��������⣺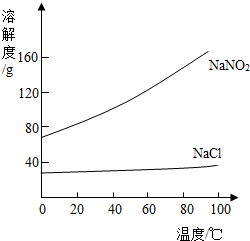 13��ͨ��һ���ѧϰ��������ѧ���Ļ�ѧ֪ʶ����������⣮
13��ͨ��һ���ѧϰ��������ѧ���Ļ�ѧ֪ʶ����������⣮