��Ŀ����
ijͬѧ��ʵ���ҷ���һ����ɫ��ĩ�������÷�ĩȡ���������Թ��У�����ϡ���ᣬ�����ݲ�����������벢�����������̽�������1��������⣺������ijɷ���ʲô��
��2����������裺�����������CO2
��3�������ʵ�飺
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��һ֧������պȡ��������ʯ��ˮ�����ڲ���������Թܿ� | ���������а�ɫ���ʲ��� | ��ѧ����ʽ�� �� ��������CO2���ɴ˿��жϸð�ɫ��ĩ�к��� �Σ� |
��4��������⣺�ð�ɫ��ĩ�ijɷ���ʲô��
��5����������裺
����1���ð�ɫ��ĩ������ȫ����ˮ��������Na2CO3��K2CO3���ѧʽ����
����2���ð�ɫ��ĩ����ȫ��������ˮ��������CaCO3�� ���ѧʽ����
��6�������ʵ�飺
��ȡ��ɫ��ĩ�������Թ��У���ע������ˮ������ĩȫ���ܽ⣮
������е������ʯ��ˮ����Һ�г��ְ�ɫ������
��7��ʵ����ۣ����� ��ȷ������˼���жϣ���ͬѧ��������ʵ���У� ������С���û�С�����Ҫ���ڢڲ��������� ��
���𰸡���������3�����ݶ�����̼���������Ʒ�Ӧ���ǣ���5�����ݰ�ɫ��ĩ���ʵ��ܽ��Է�����7������Na2CO3��K2CO3��CaCO3��BaCO3���ܽ��Կ��ǣ�
����⣺��3��������̼��ʹ����ʯ��ˮ����ǣ���Ӧ�����������ƺͶ�����̼����������̼��ƺ�ˮ����ѧ����ʽΪCa��OH��2+CO2�TCaCO3��+H2O���ɴ��жϸð�ɫ��ĩ�к���̼����
��5����ɫ��ĩ��Na2CO3��K2CO3��CaCO3��BaCO3��̼��ƺ�̼�ᱵ������ˮ
��7��ȡ��ɫ��ĩ�������Թ��У���ע������ˮ������ĩȫ���ܽ⣬˵���÷�ĩ������ˮ����ΪCaCO3��BaCO3���ڳ�����Na2CO3��K2CO3��������ˮ���Ϳ�˵������1��ȷ��û�б�Ҫ���ڢڲ���
�ʴ�Ϊ��
��3��Ca��OH��2+CO2�TCaCO3��+H2O ̼��
��5��BaCO3
��7��1 û�� ��ΪCaCO3��BaCO3���ڳ�����Na2CO3��K2CO3��������ˮ��
�����������Ҫע�ⷽ��ʽ����д������֪������������ˮ�е��ܽ��ԣ�
����⣺��3��������̼��ʹ����ʯ��ˮ����ǣ���Ӧ�����������ƺͶ�����̼����������̼��ƺ�ˮ����ѧ����ʽΪCa��OH��2+CO2�TCaCO3��+H2O���ɴ��жϸð�ɫ��ĩ�к���̼����
��5����ɫ��ĩ��Na2CO3��K2CO3��CaCO3��BaCO3��̼��ƺ�̼�ᱵ������ˮ
��7��ȡ��ɫ��ĩ�������Թ��У���ע������ˮ������ĩȫ���ܽ⣬˵���÷�ĩ������ˮ����ΪCaCO3��BaCO3���ڳ�����Na2CO3��K2CO3��������ˮ���Ϳ�˵������1��ȷ��û�б�Ҫ���ڢڲ���
�ʴ�Ϊ��
��3��Ca��OH��2+CO2�TCaCO3��+H2O ̼��
��5��BaCO3
��7��1 û�� ��ΪCaCO3��BaCO3���ڳ�����Na2CO3��K2CO3��������ˮ��
�����������Ҫע�ⷽ��ʽ����д������֪������������ˮ�е��ܽ��ԣ�

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д�
�����Ŀ
 ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ������ͼ��ʾ������Һ��Ϊ�˲ⶨ����Һ������������������ȡ��100g����Һ���������μ�������������Ϊ10%��ϡ���ᣮ�ų��������������������ϡ�����������ϵ������ͼ��ʾ��
ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ������ͼ��ʾ������Һ��Ϊ�˲ⶨ����Һ������������������ȡ��100g����Һ���������μ�������������Ϊ10%��ϡ���ᣮ�ų��������������������ϡ�����������ϵ������ͼ��ʾ��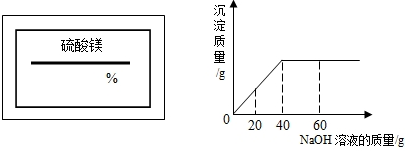
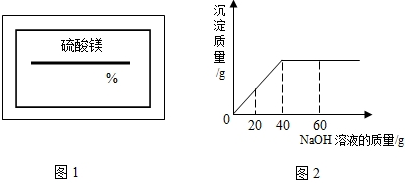
 ��2009?�Ͽ���һģ��ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ������þ��Һ��Ϊ�˲ⶨ����Һ������������������ȡ��25g����Һ����������μ���������������Ϊ10%��NaOH��Һ����Ӧ���������ɳ���������������NaOH��Һ�����Ĺ�ϵ��ͼ��ʾ������
��2009?�Ͽ���һģ��ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ������þ��Һ��Ϊ�˲ⶨ����Һ������������������ȡ��25g����Һ����������μ���������������Ϊ10%��NaOH��Һ����Ӧ���������ɳ���������������NaOH��Һ�����Ĺ�ϵ��ͼ��ʾ������ ��2008?��ǽ�����ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ����ͼ��ʾ������Һ��Ϊ�˲ⶨ����Һ������������������ȡ��20g����Һ����������μ���������������Ϊ15%��MgSO4��Һ����Ӧ���������ɳ���������������MgSO4��Һ�����Ĺ�ϵ��ͼ��ʾ����������Һ����������������
��2008?��ǽ�����ijͬѧ��ʵ���ҷ���һƿ��ǩ��ȱ����ͼ��ʾ������Һ��Ϊ�˲ⶨ����Һ������������������ȡ��20g����Һ����������μ���������������Ϊ15%��MgSO4��Һ����Ӧ���������ɳ���������������MgSO4��Һ�����Ĺ�ϵ��ͼ��ʾ����������Һ����������������