��Ŀ����
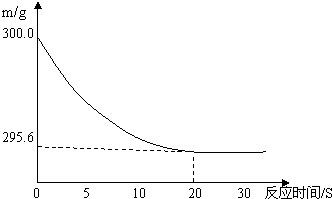
��6�֣���ʢ��22.3 g Na2CO3��NaCl����������ձ��м���216.1 gϡ����ǡ�÷�Ӧ,��Ӧ�����þ�����������ձ���ͬҩƷ������(m)�뷴Ӧʱ��(t)�Ĺ�ϵ����ͼ��ʾ���ձ���ͬҩƷ����ʼ����Ϊ300 g���ش��������⣺
(1)������������ϡ����ǡ����ȫ��Ӧʱ������ʱ��ԼΪ S��
(2)��ȫ��Ӧ����������̼�������� g��
(3)��Ӧ�õ������µIJ�������Һ��������Һ�����ʵ���������Ϊ���٣�

��6�֣�
��1��20��1�֣��� ��2��4.4 g��1�֣���
��3���⣺��μӷ�Ӧ��̼���Ƶ�����Ϊx����Ӧ���ɵ��Ȼ��Ƶ�����Ϊy
Na2CO3 + 2HCl = 2NaCl+ CO2�� + H2O
106 117 44
x y 4.4g ����1��
106/44=x/4.4g ,117/44=11.7/4.4g
��ã�x=10.6 g��y=11.7 g�� ����1��
�ձ��ﲻ������Һ������������Ϊ�� 11.7 g + (22.3 g��10.6 g) = 23.4 g��
�ձ��ﲻ������Һ������Ϊ�� 22.3 g+216.1 g��4.4 g = 234 g������1��
���ò�������Һ����������������23.4 g/ 234 g ��100%=10%������1��
����:��ͼ���п��Կ������ձ��е�������������ʱ��ʱ��Ϊ20s����Ӧǰ��Ĺ���������300-295.6=4.4�ˣ����Dz����Ķ�����̼��������������̼�ܵ�������ȥ�ˣ����Թ�����������٣���Ȼ����ݶ�����̼�������Ϳ��Լ����ˡ�

 ������ϵ�д�
������ϵ�д�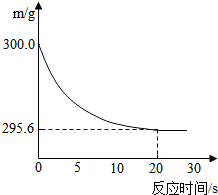 ��ʢ��22.3 g Na2CO3��NaCl����������ձ��м���216.1 gϡ����ǡ�÷�Ӧ����Ӧ�����þ�����������ձ���ͬҩƷ��������m���뷴Ӧʱ�䣨t���Ĺ�ϵ����ͼ��ʾ���ձ���ͬҩƷ����ʼ����Ϊ300 g���ش��������⣺
��ʢ��22.3 g Na2CO3��NaCl����������ձ��м���216.1 gϡ����ǡ�÷�Ӧ����Ӧ�����þ�����������ձ���ͬҩƷ��������m���뷴Ӧʱ�䣨t���Ĺ�ϵ����ͼ��ʾ���ձ���ͬҩƷ����ʼ����Ϊ300 g���ش��������⣺